
Fecal (stool) tests are used to detect colon cancer. Only very small stool samples are needed for these non-invasive cancer tests. There are several different methods currently in use.
They include:
Fecal Occult Blood Test (FOBT)
Fecal occult blood tests (FOBT) are able to detect small hidden (occult) blood present in stools, a possible sign of colon cancer or colonic polyps (precursors of cancer). Occult blood refers to very small amounts of blood that cannot be seen by visual inspection, but can be detected using chemical tests. The detection of blood in stool does not necessarily indicate cancer, it simply helps health care professionals make an accurate diagnosis. There are currently two methods used to detect occult blood, they are described below.
Guaiac Fecal Occult Blood Test (GFOBT)
The GFOBT detects the activity of peroxidase, an enzyme found in the blood. The test can be done using a home kit obtained from a health care provider. It requires a sample taken of the inside and the outside of three separate bowel movements. The samples are then placed on appropriate slides and returned for testing. Some foods, medications, and supplements can cause false positives, so be sure to carefully follow instructions from your health care provider or the test manufacturer.1, 2
Fecal Immunochemical Test (FIT)
The fecal immunochemical test is a more advanced, easier to use FOBT that has shown a better combination of sensitivity and specificity. The test uses antibodies to detect the presence of blood hemoglobin. This method is not affected by food, medication, or supplements and it is also easier for subjects to obtain an adequate sample. The FIT only requires samples from the surface of two bowel movements. Although the FIT is more user friendly, it is still important to carefully follow your health care providers instructions.1, 2
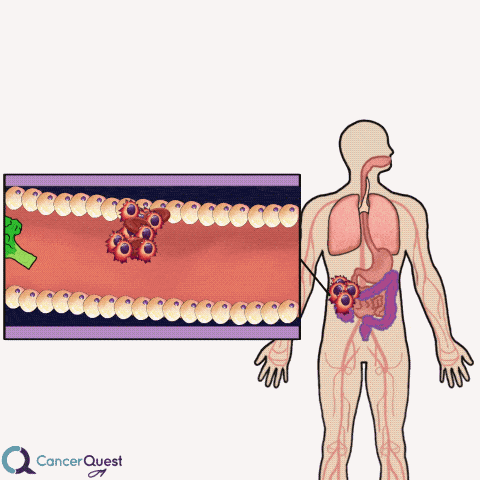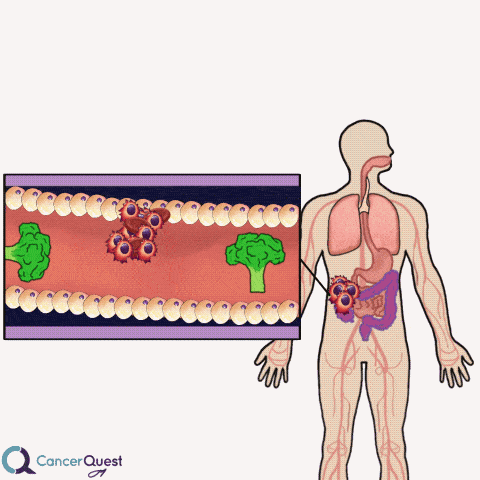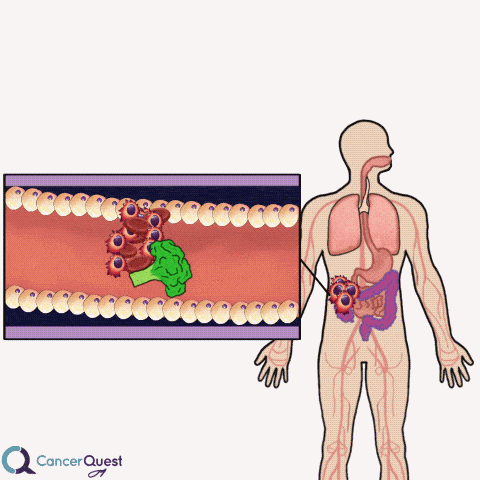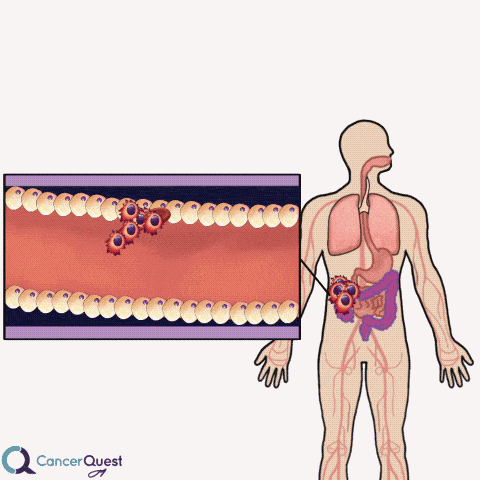
Colon cancer often forms as extensions to the colon wall. These growths can be hit by passing food and cause cancer cells and blood to be released into the colon. The cells and blood leave the body with the stool (feces).

Samples of stool can be examined for the presence of blood cells. If blood is found, it could indicate that colon or rectal cancer.
Fecal DNA Test
As part of the normal turnover that occurs in the cells that line the bowel, many of the epithelial cells that line the intestines are shed into the waste. As discussed in the section on tumor suppressors, many colon cancers share a common genetic defect in the APC gene. The cells that are shed into the fecal matter contain DNA and tests have been developed to detect mutations in the APC gene. This technique is able to detect the DNA from colon cancer cells even at early stages of cancer. Its non-invasive nature makes it a promising new screening tool.3, 4
In 2014, the FDA approved the first stool DNA test for colon cancer, Cologuard®.5, 6, 7 The test detects mutations in genes associated with colorectal cancer, and modifications (methylation) of other cancer genes. The test also detects the presence of hemoglobin (blood) in stool. Like any medical test, Cologuard® is not 100% accurate. In a large clinical trial, the test was able to detect 92% of stage 1-4 colorectal cancers.8 Because the genetic changes seen in cancer happen over time, the test can also detect pre-cancerous conditions. In the clinical trial, the test was able to detect 69% of 'high risk' pre-cancers, those thought to be most likely to progress to cancer.8
Learn more about cancer genes.
Learn more about DNA methylation in cancer.
- 1ab Guittet L et al. Comparison of a guaiac based and an immunochemical faecal occult blood test in screening for colorectal cancer in a general average risk population. Gut 2007 Feb; 56:210-4. [PUBMED]
- 2ab A Smith, GP Young, SR Cole, P Bampton. Comparison of a Brush-Sampling Fecal Immunochemical Test for Hemoglobin With a Sensitive Guaiac-Based Fecal Occult Blood Test in Detection of Colorectal Neoplasia. Cancer. 2006;107:21529. [PUBMED]
- 3 Calistri D, Rengucci C, Casadei Gardini A, Frassineti GL, Scarpi E, Zoli W, Falcini F, Silvestrini R, Amadori D. Fecal DNA for noninvasive diagnosis of colorectal cancer in immunochemical fecal occult blood test-positive individuals. Cancer Epidemiol Biomarkers Prev. 2010 Oct;19(10):2647-54. [PUBMED]
- 4 Ahlquist DA. Molecular detection of colorectal neoplasia. Gastroenterology. 2010 Jun;138(6):2127-39. [PUBMED]
- 5 FDA approval of Cologuard. http://www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm409021.htm Accessed 10-20-2016
- 6 FDA Cologuard fact sheet http://www.fda.gov/MedicalDevices/ProductsandMedicalProcedures/DeviceApprovalsandClearances/Recently-ApprovedDevices/ucm410569.htm Accessed 10-20-2016
- 7 Cologuard manufacturer's site http://www.cologuardtest.com/ Accessed 10-20-2016
- 8ab Imperiale TF, Ransohoff DF, Itzkowitz SH, Levin TR, Lavin P, Lidgard GP, Ahlquist DA, Berger BM. Multitarget stool DNA testing for colorectal-cancer screening. N Engl J Med. 2014 Apr 3;370(14):1287-97. doi: 10.1056/NEJMoa1311194. Epub 2014 Mar 19.
