Medical tests or studies are frequently used to identify medical conditions. Some tests can identify the presence of a condition before the patient exhibits symptoms. There are many different types of medical tests.
The technical name for things that are measured is a combination of the word 'biological' and 'marker' - biomarker. The term biomarker "refers to a broad subcategory of medical signs - that is, objective indications of the externally observed medical condition of the patient - which can be measured with accuracy and reproducibility."1 They include simple things like taking a pulse, listening to someone's heart, or taking their blood pressure. Some diagnostic and screening tests, such as those mentioned above, are not invasive. Others involve tests of blood or tissue taken from the patient.
The next sections contain information on some biomarkers used in medical tests, with an emphasis on those used in the diagnosis and detection of cancer.
- Types of biomarkers
- Uses of biomarkers
- Requirements for biomarkers
- Limitations of biomarkers
- Biomarkers for the detection of cancer
A biomarker is something that can be found in blood, tissue, or body fluids that can indicate a medical condition, such as disease, infection, or another abnormal condition.2 Biomarkers have quite a few uses, including monitoring patients' response to treatments and whether their disease is progressing. For cancer, many of the biomarkers are substances that are produced by tumor cells or chemicals produced in response to the presence of cancer in the body. Older examples of disease-indicating biomarkers include cancer indicators such as Bence-Jones proteins in multiple myeloma patients and carcinoembryonic antigen in colon cancer patients.3

Cancer can be detected indirectly by looking at products produced by, or induced by, the cancer. These biomarkers can be found in body fluids or excretions. Different cancers can be detected in different body products.
Biomarkers can be identified in a variety of different ways. They can be found in fluids excreted/secreted from the body, such as feces, urine or sputum. Biomarkers can be also be identified in the blood using minimally invasive tests, such as drawing blood (The Bence Jones proteins mentioned above are found in the urine, and CEA is found in the blood). Some biomarkers are found inside tissues or organs and therefore require a biopsy or imaging to evaluate them.3
Biomarkers can be discovered in a variety of different ways. Advances in technology used to sequence DNA and identify tiny amounts of proteins have provided a great deal of information on the potential of many different molecules to serve as markers for disease, including cancer.4
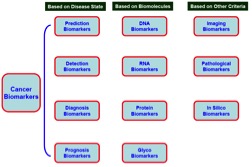
Types of Biomarkers
In theory, there are many biological molecules that can serve as indicators of disease. All molecules found in the body that have been altered in some way by disease have the potential to serve as a biomarker. Changes can be changes in the marker structure (called qualitative changes) or in the marker amount (called quantitative changes). Some biomarkers are described below:
Deoxyribonucleic acid (DNA), the carrier of our genetic code, is widely used as a biomarker. Mutations found in proto-oncogenes, which lead to oncogene formation, are commonly used to evaluate cancer. Tumor-suppressors, genes that aid in cancer prevention, can also be monitored as biomarkers. Other abnormal genetic changes, such as the number of copies of a particular gene and the fusion of genes that are not normally found together (via translocations) are also used as biomarkers. 3 The source of DNA can be tissue, serum, sputum, saliva, cerebrospinal fluid, or circulating tumor cells (CTCs).
Ribonucleic acid (RNA) is very similar to DNA, and changes in RNA may also indicate disease. Small, noncoding RNA molecules, known as microRNAs (miRNAs) are the basis of a growing field of cancer and biomarker research. miRNAs have been identified as markers for many different types of cancer, including colorectal, breast, leukemia, liver, lung, and pancreatic cancer. 5
Proteins are arguably the most important type of biomarker to date. They are responsible for controlling most cellular processes. Proteins may be a more reliable indicator of some conditions because they are created and destroyed rapidly, unlike DNA and some RNA molecules. Proteins can give a real-time indication of the body's condition. 3
Viruses can also serve as biomarkers. Viral infections contribute to about 15-20% of all cancers. For example, Epstein-Barr virus (EBV) has been linked to carcinoma and lymphoma. Human papillomavirus (HPV) has also been showed to be the major cause of cervical cancer and also causes a growing percentage of head and neck cancers. In April of 2014, the FDA approved a HPV DNA test as a primary cervical cancer screening method. 6
Bacteria can also be used as a biomarker. Bacteria that cause chronic, low-level inflammation have been known to cause cancer. For example, H. pylori, a bacterium that inhabits the stomach lining has been linked to the formation of ulcers and stomach cancer. 3
Exosomes are small bubble-shaped structures (vesicles) secreted by cells. They can contain DNA, RNA, miRNA, and/or proteins specific to cancer cells. 7. Exosomes can be found in bodily fluids, such as blood and urine. In many cancers, exosomes have been found to prepare distant sites for metastasis. There are many ongoing efforts examining the use of exosomes as a tool for the diagnosis of cancer and predicting likely cancer outcomes. 8
Cancer cells themselves can be used as biomarkers. Cancer cells circulating the blood stream, known as circulating tumor cells (CTCs), can also be used to provide an indication of disease progression. 9 Cancer stem cells (CSCs) have also been used to monitor disease. CSCs are thought to be a small subset of tumor cells that actively drive tumor growth and spread. 10
Uses of Biomarkers
Biomarkers have several main uses in cancer.
- Determining what condition a patient has (diagnosis): The goal of using biomarkers as a diagnostic tool is to identify cancer in its early stages. Diagnostic tests can be used to determine the exact nature of a cancer. They may be used to help guide treatment plans.
- Determining the likely outcome for the patient (prognosis): A prognostic biomarker is used to determine how aggressive the cancer likely to be.
- Predicting disease progression: A predictive biomarker is used to assess how well a patient will respond to a particular treatment. 3 This type of biomarker can also be used to determine what drugs or chemotherapy agents will be most effective for a particular patient. For example, the drugs erlotinib and getfitinib used to treat lung cancer are only effective in patients with a specific mutation in a cell surface protein - called EGFR. 3
- Monitoring disease progression: Biomarkers can be used to track how well a treatment is working and guide future treatment decisions.
Biomarker Requirements
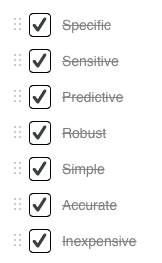
Despite the fact that many molecules have potential to serve as biomarkers, few have made it to the clinic and gained approval from the U.S. Food and Drug Administration (FDA). To be effective, a biomarker must be able to diagnose early stage tumors and be highly specific to the condition of interest. The molecule must also be present at high enough levels to be detectable. Ideally, the biomarker should be readily found in the blood, serum or other body fluid of affected individuals. Other features include low testing cost, and tests that are easily done. 11, 12
Biomarker Limitations
Although biomarkers have been successfully used to identify and monitor disease, there are trade-offs between sensitivity, specificity, and cost. 13 Sensitivity is the ability of a test to identify people who are affected by the condition. Specificity is the ability of a test to exclude those who do not have the condition.
Learn more about sensitivity and specificity
Another limitation of biomarkers is the possibility of over-diagnosis. Biomarkers may identify cancer that may never have led to symptoms during a person's lifetime. 13 An example of this is prostate specific antigen (PSA), a biomarker for prostate cancer. PSA tests are not able to distinguish between slow growing (indolent) cancers and aggressive cancers that would harm the patient. Because of this limitation, the test is no longer recommended for screening of men at normal risk for prostate cancer. 14
Approved Cancer Biomarkers
List of FDA-approved cancer biomarkers 11
- AFP (Alpha-Fetoprotein): elevated in non-seminomatous germ-cell tumors (NSGCTs) and liver cancer (also called hepatocellular carcinoma or HCC)
- CA-125 (Cancer Antigen 125): used to monitor response to treatment and recurrence of ovarian cancer
- HE4 (Human Epididymis Protein 4): used to monitor recurrence of ovarian cancer
- TG (Thyroglobulin): a marker for thyroid cancer
- PSA (Prostate-Specific Antigen): increased levels in men may indicate prostate cancer
- CEA (Carcinoembryonic Antigen): a marker for colon cancer
- CA 19-9 (Cancer Antigen 19-9): a marker used to measure response to treatment in pancreatic cancer
- CA 15-3/CA 27-29 (Cancer Antigen 15-3/Cancer Antigen 27-29): used to monitor breast cancer
- HER-2/neu (Human Epidermal Growth Factor Receptor 2/neu): predictive marker for breast cancer
- 1 Kyle Strimbu and Jorge A. Tavel, MD. What are Biomarkers? Curr Opin HIV AIDS. 2010 Nov; 5(6): 463–466. [<a href=“https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3078627/” target=“_blank”>PubMedCentral Article</a>]
- 2 NCI Dictionary of Cancer Terms: biomarker. National Cancer Institute. National Institutes of Health. U.S. Department of Health and Human Services. USA.gov. [http://www.cancer.gov/publications/dictionaries/cancer-terms?cdrid=45618]
- 3abcdefg Mishra A, Verma M. Cancers (Basel). 2010 Mar 22;2(1):190-208. doi: 10.3390/cancers2010190. Cancer biomarkers: are we ready for the prime time? [PUBMED]
- 4 Tardif JC, Heinonen T, Orloff D, Libby P. Circulation. 2006 Jun 27;113(25):2936-42. Vascular biomarkers and surrogates in cardiovascular disease. [PUBMED]
- 5 Bartels CL, Tsongalis GJ. MicroRNAs: novel biomarkers for human cancer. Clin Chem. 2009 Apr;55(4):623-31. doi: 10.1373/clinchem.2008.112805. Epub 2009 Feb 26. [PUBMED]
- 6 FDA approves first human papillomavirus test for primary cervical cancer screening. U.S. Food and Drug Administration. [http://www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm394773.htm]
- 7 Thakur BK, Zhang H, Becker A, Matei I, Huang Y, Costa-Silva B, Zheng Y, Hoshino A, Brazier H, Xiang J, Williams C, Rodriguez-Barrueco R, Silva JM, Zhang W, Hearn S, Elemento O, Paknejad N, Manova-Todorova K, Welte K, Bromberg J, Peinado H, Lyden D. Double-stranded DNA in exosomes: a novel biomarker in cancer detection. Cell Res. 2014 Jun;24(6):766-9. doi: 10.1038/cr.2014.44. Epub 2014 Apr 8. [PUBMED]
- 8 Tickner JA, Urquhart AJ, Stephenson SA, Richard DJ, O'Byrne KJ. Functions and therapeutic roles of exosomes in cancer. Front Oncol. 2014 May 27;4:127. doi: 10.3389/fonc.2014.00127. eCollection 2014. [PUBMED]
- 9 Turner N, Pestrin M, Galardi F, De Luca F, Malorni L, Di Leo A. Can biomarker assessment on circulating tumor cells help direct therapy in metastatic breast cancer? Cancers (Basel). 2014 Mar 25;6(2):684-707. doi: 10.3390/cancers6020684. [PUBMED]
- 10 Kreso A, Dick JE. Evolution of the cancer stem cell model. Cell Stem Cell. 2014 Mar 6;14(3):275-91. doi: 10.1016/j.stem.2014.02.006. [PUBMED]
- 11ab Rhea JM, Molinaro RJ. Cancer biomarkers: surviving the journey from bench to bedside. MLO Med Lab Obs. 2011 Mar;43(3):10-2, 16, 18; quiz 20, 22. [PUBMED]
- 12 Karley D, Gupta D, Tiwari A. Biomarker for cancer: a great promise for future. World J Oncol. 2011 Jul;2(4):151-157.
- 13ab Hartwell L, Mankoff D, Paulovich A, Ramsey S, Swisher E. Cancer biomarkers: a systems approach. Nat Biotechnol. 2006 Aug;24(8):905-8. [PUBMED]
- 14 Detection of Prostate Cancer. American Urological Association. [https://www.auanet.org/education/guidelines/prostate-cancer-detection.cfm]
