Breast cancer ranks as one of the leading cancer types in the number of new cases diagnosed and is second only to lung cancer as the the most prevalent cause of cancer death in women. In 2023, the American Cancer Society estimates approximately 300,590 new cases of breast cancer will be diagnosed and 43,700 breast cancer deaths. Although it is much less common, it is important to note that men also develop breast cancer. Approximately 530 of the estimated deaths due to breast cancer in 2023 will be men.1
Young women DO get cancer. Watch an interview with Robin Shoulla, a breast cancer survivor diagnosed when she was 17 years old.
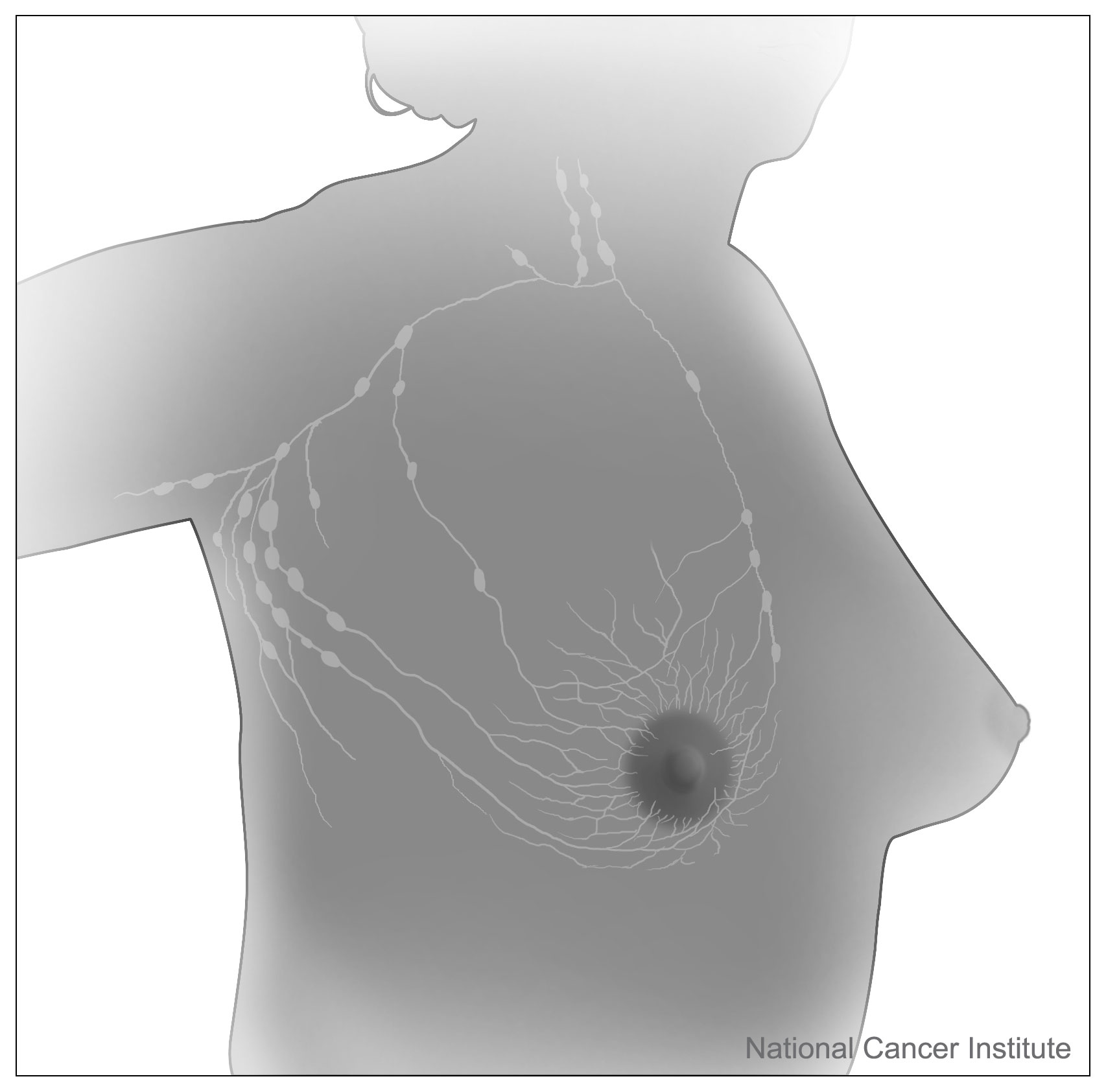
Anatomy Of The Breast
In women, the breasts are made up of milk-producing glands (lobules), milk ducts, and connective tissue (stroma). Milk is produced by cells in the lobules and moves from these sacs, through the mammary ducts, to the nipple. Most breast cancers originate in mammary ducts.2


Blood and lymphatic vessels are found within the stroma surrounding the lobules and ducts:
- Blood vessels are part of the circulatory system. They supply oxygen and nutrients to and remove waste from the cells of the breast.
- Lymphatic vessels are part of a large network termed the lymphatic system. These vessels collect and carry fluid and cells from the tissues of the body. Smaller lymphatic vessels merge with larger ones, as streams merge into a river. Large vessels empty into grape-like clusters of lymphatic tissue called lymph nodes. The lymphatic vessels in the breast carry lymphatic fluid to a mass of lymph nodes located near the underarm.
Types
There are several different kinds of breast cancer. However, the majority of breast cancer cases are classified as either in situ or invasive. Both cancer types are described below.
Carcinoma In Situ
Lobular Carcinoma In Situ (LCIS)
Lobular carcinoma in situ describes a non-invasive pre-cancerous condition that is confined to the milk-producing glands (lobules) of the breast. Tumors classified as LCIS are made up of small cells that are all the same and do not leave the lobules. Since LCIS does not leave the breast lobules, it increases the risk of developing invase cancer but is not yet classified as invasive and is therefore, technically, not cancer. It is also referred to as lobular neoplasia (LN), which is a more accurate name because LCIS/LN is only a precursor to cancer. LN also includes atypical lobular hyperplasia (ALH), another abnormal breast finding in which there are too many cells. 3
Lobular carcinoma in situ is most commonly found in pre-menopausal women between the ages 40 and 50 4. It is not usually found on a mammogram and generally does not produce a lump 4. These tumors are usually HER2 negative (-) and ER/PR positive (+), which simply means that the cancer cells may still rely on estrogen hormone for growth signals. Therefore, treating the cells with a hormone therapy (tamoxifen), may stop the cancer cells from dividing. Overall LCIS is highly treatable and in many cases, continued observation is enough.
Ductal Carcinoma In Situ (DCIS)
Ductal carcinoma in situ, or “Stage 0 cancer”, describes a non-invasive or pre-invasive breast condition in which abnormal cells are found in the lining of breast milk ducts. These abnormal cells have not spread/invaded beyond the walls of the ducts into surrounding breast tissue. DCIS is not cancer. It is referred to as “pre-invasive” because some cases will progress into cancers, while others will not. Currently, there is no effective way to determine this.5, 6
Unlike LCIS, DCIS can be detected on a mammogram and usually does produce a lump, although the lump may be too small to feel or in a location that is not easily felt. This is because excess calcium is produced and the solid calcium shows up on scans. DCIS accounts for about 85% of in situ cancers diagnosed each year in the United States. Most cases of DCIS (~98%) will not become metastatic itself, but around half of all cases progress to invasive breast cancer (IBC). The progression of DCIS to IBC is not completely understood and treatment recommendations tend to vary.
There are several treatment options for DCIS. They include:
1. Watching the growth without treating it.
2. Surgery - There are 2 major surgical treatment categories for DCIS: breast conserving and breast removing. The most common treatment plan for DCIS is breast-conserving surgery.
3. Radiation - This is often combined with surgery.
4. Hormonal treaments - These treatments are designed to starve the cells of growth signals.
There is an ongoing debate among healthcare professionals as to what the best DCIS treatment option is. Some doctors believe those with DCIS should receive aggressive treatment since there is a possibility that the DCIS may turn into an invasive cancer. Other doctors regard aggressive DCIS treatment as over-treatment and prefer to monitor and wait.7, 8, 9
Furthermore, recurrence of DCIS is a possibility; prognostic factors are calculated based on nuclear grade (most important factor), cell necrosis (premature cell death), and cell and tumor architecture. 50-75% of DCIS lesions are ER/PR + and 30-50% of the time (more often in high-grade lesions) HER2 is over-expressed. DCIS cases that overexpress HER-2 are associated with progressive cancer. These biological markers cannot fully predict recurrence risk, but may be helpful in follow up observation and treatment planning.10
Invasive Breast Carcinoma (IBC)
Images of invasive breast cancer: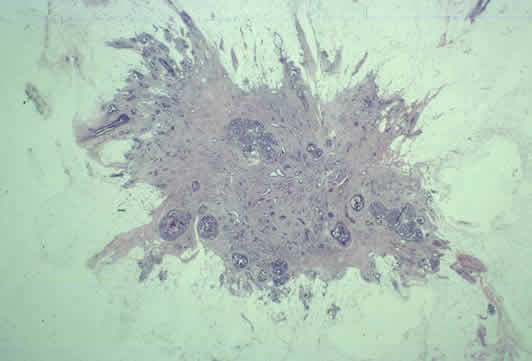
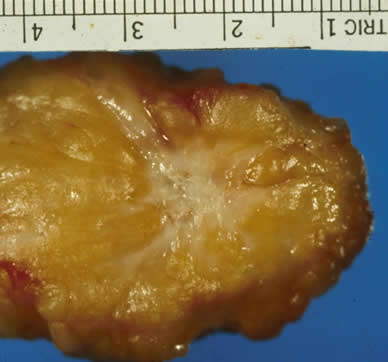
Left: Pathology slide image of cancerous breast tissue, Right: Tumor (white area) in fatty breast tissue.
Images courtesy: C. Whitaker Sewell, MD - Professor of Pathology, Emory University School of Medicine
Invasive Lobular Carcinoma (ILC)
Invasive lobular carcinoma develops in the milk-producing glands (lobules) of the breast. ILC has the ability to spread to other parts of the body, (most commonly bone, brain, liver, and lungs) either through the bloodstream or the lymphatic system. ILC usually presents as an abnormal feeling breast (most often a thickening) and not as a hard mass that can be felt. ILC is less likely to show up on a mammogram than invasive ductal carcinoma.
Women over the age of 40 have an increased risk of developing invasive lobular carcinoma, with most cases occurring in women between the ages 45-56.
Invasive Ductal Carcinoma (IDC)
Invasive ductal carcinoma is the most common type of invasive breast cancer, responsible for almost 85% of cases. IDC starts in the milk ducts and invades the surrounding tissue. IDC also has the ability to move to other parts of the body (most commonly bone, brain, liver, and lungs) either through the bloodstream or the lymphatic system. IDC develops as a hard lump with irregular borders that usually shows up as a spiked mass on a mammogram.
Women over the age of 40 have an increased risk of developing invasive ductal carcinoma, with around 50% of cases occurring in women over the age of 65.
Risk Factors
Factors that influence risk of developing breast cancer include:
- Prior History of Breast Disease
- Family History of Breast Disease
- Age
- Race
- Reproductive and Menstrual History
- Radiation Exposure
- Dietary Factors
The relative effects of these and other risk factors in any given case of cancer is variable and very difficult to determine with accuracy at this time. Some of these and other risk factors are discussed in the following sections.
Prior History of Breast Disease
History of breast cancer, regardless of the type, greatly increases an individual's risk for future development of breast cancer. The second occurrence is most likely to be classified as invasive and ductal, rather than a lobular breast cancer.11 Such women are strongly advised to carefully monitor themselves and receive mammograms on a regular basis.
Family History of Breast Disease
It is possible to inherit defective genes that lead to the development of a familial form of a particular cancer type. Individuals with a family history of breast cancer are therefore at an increased risk of developing the disease. The degree of risk depends upon the type of relative affected. For example, risk is higher if an immediate family member has been diagnosed with breast cancer. The more closely related an individual is to someone with breast cancer, the more likely they will share the same genes that increased the risk of the affected individual. Risk also increases with the number of relatives affected.11
Age
Breast cancer risk is low before age 30 and increases with age, leveling off at the age of 80.11 More information about the relationship between cancer and age can be found in the Cancer Biology section.
Race
African American women are more likely to be diagnosed with early-onset (before age 45) breast cancer compared to white American women. This trend in incidence shifts, however, for women in their fifties. When comparing overall lifetime risk, African American women have a lower risk of developing breast cancer compared to white American women.12
Cancer survival rates are greatly affected by the stage at which a cancer is detected. Early-onset cancers are generally more aggressive than cancers that develop later in life. Studies show that African American women have a greater chance of being diagnosed with high-grade and estrogen receptor (ER)-negative tumors. Regardless of their lower overall lifetime risk of developing the disease, African American women have a significantly higher chance of dying from breast cancer than any other ethnic group in the United States.13
Reasons for these differences in cancer occurrence and death rate are still unclear. One explanation for the disparity is the socioeconomic differences that exist between ethnic groups. In this view, limited access to health care and clinical trials and the high cost of treatments may account for the higher African American breast cancer death rate. New investigations focused on biological differences, however, suggest that African Americans may develop tumors that are harder to treat. Current research seems to suggest that the disparity in cancer death rate is due to a combination of socioeconomic and genetic factors.13 Clearly, more work needs to be done in this area.
Reproductive and Menstrual History
Exposure to estrogen is associated with increased breast cancer risk. For this reason, women who experience menarche (begin their menstrual cycle) before age 12 and the onset of menopause at age 55 or older are at an increased risk of developing breast cancer.11
On the other hand, women who carry pregnancy to full term at a young age have a decreased risk of developing breast cancer. During pregnancy, maternal hormone levels change drastically. Studies show that some of these hormones may provide anti-estrogen effects, protecting individuals from the negative effects of estrogen. Examples of proteins thought to impact cancer risk include:
- Alpha-fetoprotein: Alpha-fetoprotein is a glycoprotein produced by the liver of a developing fetus. Since the protein is at its highest level during the third trimester, a pregnancy that is not carried to full term may not provide a protective effect.14
- Human Chorionic Gonadotropin (hCG): Human chorionic gonadotropin is a hormone produced by the placenta during pregnancy. The protein is elevated during the first trimester of pregnancy. Experiments with breast cancer cells suggest that the effects of hCG may partially explain the higher rate of breast cancer among nulliparous (non-child bearing) women.15
Hormone Replacement Therapy
Hormone replacement therapy (HRT) is often prescribed to control menopausal symptoms that include decreased bone density, sexual dysfunction, fatigue, and mood swings. HRT may include treatment with estrogen alone or a combination of both estrogen and progesterone. Studies indicate that use of menopausal hormone therapy, currently or within the past five years, is associated with an increased risk of developing breast cancer.16 Several studies have shown that individuals who have stopped using HRT for more than five years are not at a significantly greater risk.11 Much of the information regarding HRT and breast cancer risks has come from population studies that may be difficult to compare with each other. The actual impact of HRT on breast cancer risk is still under investigation.
Exposure to Diethylstilbestrol
Diethylstilbestrol (DES) is a man-made estrogen that was prescribed between 1947 and 1971 to treat complications associated with pregnancy. Women who ingested this chemical during their pregnancy are at a slightly increased risk of developing breast cancer. Female offspring of women who were exposed to DES have a higher incidence of reproductive problems and cancers of the vagina and cervix.17
Radiation Exposure
Exposure to radiation, such as radiation therapy used to treat Hodgkin lymphoma, increases risk of breast cancer throughout the remainder of a woman's lifetime.11Relative risk depends on the age at which irradiation occurred, time since treatment, and radiation dose received. Women irradiated at a young age (before age of 30) are at an increased risk of developing breast cancer later in life compared to older women. Advances in therapeutic radiation techniques have resulted in the use of lower doses of radiation and exposure of smaller regions of the body. Such advances will hopefully reduce breast cancer risk due to irradiation therapy in years to come.18
Dietary Factors
It is very difficult to identify dietary items that cause a particular cancer. Factors that may increase breast cancer risk include high fat intake, high alcohol consumption, and a diet rich in overcooked meats.
- Overcooked Meats: Heterocyclic amines are chemicals formed in the cooking process of meat products such as fish, beef, pork, and chicken. Exposure to high amounts of these chemicals, caused by a diet rich in broiled, grilled, or fried meat, has been correlated with increased risk of breast cancer in women.19
- Alcohol Consumption : According to a 2016 report by American Institute for Cancer Research, even consuming just one glass of wine or other alcoholic beverage per day can significantly increase breast cancer risk (pre-menopausal breast cancer risk by 5 percent and post-menopausal breast cancer risk by 9 percent). This report comprises results and information from 119 studies, which includes data on 12 million women and 260,000 individual cases of breast cancer. In addition to stating the risks that come with alcohol consumption, the report states that exercise can lower breast cancer risk, as well as mitigate the effects of the alcohol.20
Symptoms
Symptoms
The American Cancer Society lists the following symptoms associated with breast cancer:
- Presence of a lump or thickening in the breast;
- Swelling, dimpling, redness, or soreness of skin;
- Change in shape or appearance of the nipple; and
- Nipple discharge.
A physician should be consulted if they persist. It is important to note, however, that these symptoms may be caused by factors unrelated to cancer. Most breast cancers are detected on mammograms before any symptoms appear.21 Therefore, regular screening is of the utmost importance.
Detection And Diagnosis
It is estimated that in the United States 234,190 cases of breast cancer will be diagnosed and 40,730 people will die from this disease in 2015. Breast cancer is the most commonly diagnosed cancer among women. Despite these large numbers, the death rates from breast cancer have been steadily falling since 1990. 22The decrease in mortality is partly due to early detection methods, advanced diagnostic techniques, and improved treatment.
The three major ways to screen for breast cancer are self examination, clinical examination, and mammography. Since no one method is 100% effective, it is suggested that all three be used regularly. The American Cancer Society has published guidelines for screening for women with a normal risk for breast cancer.23, 24, 25 Women at high risk due to inherited mutations are suggested to follow a different set of guidelines.26Learn more about the benefits of early breast cancer detection.
Advances in breast cancer treatment are facilitated by the knowledge gained from tumor biopsy and pathology. These techniques allow doctors to develop individual treatments for each patient based on their distinctive tumor biology. New techniques are constantly being researched to improve the detection, diagnosis, and treatment of breast cancer.
Watch the video to hear what breast cancer survivor Monica Pearson thinks about mammograms and breast cancer detection. Watch the full interview with Monica Pearson.
Below is a list of tests used in breast cancer detection and diagnosis: Detection Diagnosis One of the new breast cancer detection techniques being studied is:
|
Pathology Report And Staging
The Pathology Report
If there is suspicion that a patient may have breast cancer, a sample of tissue (biopsy) may be taken for examination. After a biopsy is taken, the physician who performed the biopsy sends the specimen to a pathologist. The pathologist examines the specimens at both the macroscopic (visible with the naked eye) and microscopic (requiring magnification) levels and then sends a pathology report to the physician. The report contains information about the tissue's appearance, cellular make up, and state of disease or normalcy. For more information about the pathology report, refer to the Diagnosis & Detection section.
Staging
Staging a cancer is a way of describing the extent of the disease. One of the most common methods used for breast cancer staging is called the T/N/M system, which assigns a degree of severity based on the size, location, and spread of cancer in the body. Details of the T/N/M system and a breast cancer staging chart can be found in the Diagnosis and Detection section. Click on the following links to for more information:
Tumor Biology
Genetic changes that occur in cancer include mutation of key regulatory genes, changes in protein products, and changes in the amount of product produced by genes (gene expression). As changes accumulate, cells become more abnormal and cancer progresses. Details of genetic change associated with cancer can be found in the mutation section. Some of the genetic elements that have been shown to be important in the development of breast cancer are listed and discussed below:
- BRCA1 and BRCA2 Genes
- HER-2/neu Gene
- Estrogen Receptor (ER)
- PTEN Gene and Cowden Syndrome
- TP53 Gene and Li-Fraumeni Syndrome
- ATM Gene and Ataxia-Telangiectasia
BRCA1 and BRCA2
The BRCA1 and BRCA2 genes are important tumor suppressor genes associated with the repair of damaged DNA. Information about BRCA1 and BRCA2 can be found in the Cancer Genes section.
HER-2/neu
HER-2/neu is an oncogene that is amplified in up to 30% of invasive breast cancers.27Information about oncogenes, gene amplification, and HER-2/neu can be found in the Cancer Genes section:
Learn more about oncogenes
Learn more about gene amplification
Learn more about HER-2/neu
Estrogen Receptor (ER)
The estrogen receptor (ER) is a protein that binds to estrogen that enters the cell. Estrogen is a steroid (lipid) hormone produced by the ovaries. The combination of protein and hormone then acts as a transcription factor to turn on genes that enable the target cells to divide. The receptor is active in the cells of the female reproductive organs, such as breasts and ovaries.
The mechanism of estrogen action is shown below.
The small green ball represents estrogen. It is a small hydrophobic molecule and it enters cells by crossing through the lipid membrane. Once in the cell, the estrogen binds to its receptor (colored orange) and the complex binds to DNA in the nucleus causing genes to be transcribed.
Several drugs have been developed to try to block the gene-activating function of estrogen. A commonly prescribed example is tamoxifen, a drug that partially inhibits the activity of estrogen. Tamoxifen is colored pink in the animation below.
These drugs should slow the growth of cancers that are growing in response to the presence of estrogen and its receptor. More information on estrogen receptors and cancer can be found in the section on cancer treatments.
Cowden Syndrome and the PTEN gene
Cowden's syndrome (CS), also known as multiple hamartoma syndrome, accounts for a small fraction (less than 1%) of hereditary breast cancers. This syndrome is caused by mutation in PTEN, a gene that functions as a tumor suppressor and helps to control the cell cycle and regulate apoptosis.
CS is also associated with abnormalities of the thyroid, gastrointestinal tract, and central nervous system.28 Women with CS have between a 25%-50% risk of developing breast cancer.29More information about tumor suppressors and the cell cycle can be found in the Cancer Genes and Cell Division sections.
Learn more about tumor suppressors
Learn more about the cell cycle
TP53 Gene and Li-Fraumeni Syndrome
Li-Fraumeni syndrome (LFS) accounts for a small fraction (less than 1%) of all breast cancer cases. This syndrome is caused by mutation in TP53 (also referred to as p53), an important tumor suppressor gene. LFS is associated with diagnosis of breast carcinoma at an early age (before age 40), as well as sarcomas, brain tumors, leukemias, and adrenocortical carcinomas.29
Learn more about abnormal p53 and cancer development
ATM Gene and Ataxia-Telangiectasia
Ataxia-telangectasia (AT) is a disorder caused by mutation in ATM, a tumor suppressor gene that helps to detect and repair damaged DNA.30 AT is associated with neurological (nervous system) deterioration, telangiectasias (red dots, often on the face, caused by blood vessels visible through the skin), immune deficiency, and hypersensitivity to ionizing radiation.29
As described in the Breast Cancer: Risk Factors, radiation exposure is a risk factor for breast cancer. Hypersensitivity to ionizing radiation caused by the ATM mutation therefore increases one's risk of developing breast cancer.
More Info on BRCA
BRCA1 and BRCA2 consist of about 17,000 bases each. This long coding sequence can make it difficult to find a single, function-altering mutation. The BRCA testing process has been separated into two stages in order to limit the complexity of the process. In the first stage, blood is drawn from the patient affected with ovarian or breast cancer. The DNA from the white blood cells is then extracted and analyzed in a laboratory to identify any BRCA mutations. In the second stage, blood is drawn from the affected patient's family members. Instead of searching through the entire gene of the family members, researchers look for the same BRCA mutation they found in the affected patient.31
BRCA mutations are responsible for 90% of hereditary ovarian cancer as well as 84% of hereditary breast cancer32. A BRCA2 mutation in men also raises the risk for male breast cancer31. These numbers may seem high, but BRCA mutations actually are uncommon in the general population. One of the highest frequencies (2%) of BRCA mutations occurs in the Ashkenazi Jewish population.
Genetic Testing for BRCA
Genetic testing can be intimidating. Genetic counselors are trained healthcare providers who help with the genetic testing process. They consult with patients in order to determine if a test is necessary and they interpret the data from a genetic test in order to better assess personal risk. They also help with the emotional and psychological impact that comes with genetic testing. Informed consent from the patient is required before any genetic testing.
Options after finding a BRCA mutation
Patients that test positive for BRCA mutation are encouraged to follow the National Comprehensive Cancer Network guidelines for breast cancer risk reduction: monthly breast self-examination, clinical breast examination semi-annually, and annual mammography and breast MRI. A prophylactic mastectomy (90-95% risk reduction) and tamoxifen (50% reduction over 5 years) are other options to reduce breast cancer risk.
Since a BRCA mutation also raises the risk of developing ovarian cancer, steps can be taken in order to reduce that risk. A prophylactic salpingo-oophorectomy (surgery that removes the ovaries) reduces ovarian cancer risk by 98% and oral contraceptives reduce risk 60% over 6 years use. If prophylactic surgery is not wanted: Semi-annual pelvic exams with transvaginal ultrasound with color Doppler should begin at 35 years of age.32
Human Epidemal Growth Factor 2 (HER2)
HER2 tumors tend to be more aggressive, so accurate identification by a pathologist is important in determining clinical treatment. Also, studies have found that ER and HER2 interaction may lead to tamoxifen resistance. This resistance is partly due to HER2's ability to inhibit the apoptotic effects of tamoxifen. Surprisingly, HER2/EGFR signaling has been suggested to be directly involved in tamoxifen growth promotion. These factors lead to the association of tamoxifen failure with HER2 overexpression.33
Progesterone Receptor (PR)
Progesterone regulates cell growth in normal breast tissue and in the uterus. PR plays an important role in mammary growth and development, especially during pregnanacy. PgR can up-regulate the expression of various genes involved in cell proliferation, survival, and tumor progression. PR is a predictive factor for the outcome of endocrine therapy. PR negative tumors have more aggressive features: they are larger, have more nodal metastases, are more likely to be aneuploidy and are more rapidly proliferating. Also, PgR negative tumors are associated with a significantly higher frequency of HER2 overexpression.32
Estrogen Receptor (ER)
Recent research has indicated there are two estrogen receptors: ERα and ER². ER²'s role in breast tissue is unclear. Studies are investigating its role in breast cancer and some conclusions and suggestions have been made. A general consensus defines tumors as either ERα+/ER²+ or ERα-/ER²+. A recent study suggested that increased ER² expression was associated with a high S-phase fraction only within ERα negative patients. Data from another study suggest that ER² may be related to proliferation in breast cancer. Overall, a consistent conclusion from studies is that ERα-/ERbeta + breast tumors are positively associated with markers suggesting a more aggressive disease.34
Treatment
The treatment of breast cancer can be broken down into two categories: early stage and advanced stage. Early stage treatment involves tumors that are confined to the breast. Advanced stage treatment involves tumors that have spread beyond the breast to other regions of the body. This spread is known as metastasis. Treatment options are dependent upon size of tumor, location, physical condition of patient, and stage of cancer.
As our focus is on the biology of the cancers and their treatments, we do not give detailed treatment guidelines. Instead, we link to organizations in the U.S. that generate the treatment guidelines.
The National Comprehensive Cancer Network (NCCN) lists the following treatments for breast cancer:
For more information about how other cancer treatments work, refer to the Cancer Treatments section.
Watch interviews from breast cancer survivors: Karen Neely Robin Shoulla Monica Pearson
Learn more about breast cancer treatment at the Winship Cancer Institute of Emory University.
Information about clinical trials:
- General clinical trial information from CancerQuest
- Click here for information about clinical trials from the National Cancer Institute.
- Click here for information about clinical trials from Georgia Clinical Trials Online.
- Click here for information about clinical trials at the Winship Cancer Institute of Emory University
Prophylactic Mastectomy, BCT, And Axillary Lymph Node Removal
Prophylactic Mastectomy
A prophylactic mastectomy can be carried out for persons at risk for developing breast cancer. An estimated 4-7% of all breast cancers are hereditary. The most well known inherited gene mutation in BRCA1 and BRCA2 significantly increases the lifetime risk for breast cancer. A prophylactic mastectomy is the most effective method of risk reduction in high risk women. The risk of newly formed breast cancer after a prophylactic mastectomy is small.35
Breast-conserving Operation (also called Breast Conservation Therapy or BCT) vs. Mastectomy
An alternative to a radical mastectomy for early stage breast cancer is breast conservation therapy. Breast conservation therapy includes lumpectomy, axillary lymph node dissection, and, usually, radiation therapy. The therapy has two goals: 1) to reduce the risk of cancer reoccurrence, and 2) to preserve the breast anatomy as best as possible.
A 22 year study of breast cancer survivors compared mastectomy to breast conservation therapy. All women in the study had been diagnosed with stage I or II breast carcinoma. Both overall survival and disease-free survival showed little difference between the two groups.36
Axillary Lymph Node Dissection
Axillary lymph node dissection is carried out with breast-conserving therapy for early stage breast cancers. It is the current standard of surgical care to reduce morbidity of axillary recurrence. The purpose of this procedure is to eliminate any cancer cells that may be residing in these lymph nodes. The risks for this procedure include infection, cutaneous numbness, long-term postoperative dysthesias and significant lymphedema.37
Breast Cancer In Men
Although it is not common, breast cancer does occur in men. In 2016 it is estimated that 2,600 new cases in males will be diagnosed with breast cancer, and 440 men will die of the disease. It tends to occur at a later age in men than in women, with the male peak age being 71 years old as opposed to peaks at 51 and 71 years old in women.38, 39, 40
- Painless lump in the breast (over 75% of cases)
- Nipple retraction, discharge, or ulceration
- Painful lump in the breast
- Paget's Disease (only about 1% of cases): This is a rare type of cancer that can appear as a skin rash (dermatitis) around the nipple. Note that this is NOT related to Paget's disease of the bone.
Detection and Diagnosis
Diagnosis of breast cancer in men is generally delayed 6 - 10 months (the time from the onset of symptoms until the patient seeks treatment). This delay is partially due to
a) the rarity of the disease
b) a lack of awareness of (and suspicion of) the disease by patients and their doctors.
Because of this delay, men present with the disease at a later stage than women:
- Stage I - ~40%
- Stage II - ~20%
- Stage III/IV - Over 40%
When symptoms are present, doctors generally use the Triple Test (TT) method to determine stage and treatment plan. The TT is the use of clinical exam, mammography or ultrasound, and fine needle aspiration (FNA) or core needle biopsy (CNB). 39, 38Watch a video about mammography, ultrasound, and/or biopsy.
Mammography has been shown to have a sensitivity of 92% and a specificity of 90% in male breast cancer, but is not used as a screening tool. 38Watch a video about sensitivity and specificity of medical tests.
Pathology
Male breast tissue is mostly fat and the lobules needed to form milk are undeveloped. Over 90% of male breast tumors are invasive (infiltrating) ductal carcinoma and ER positive.38, 39
- Obesity
- Alcohol consumption
- Liver damage or dysfunction
- Testicular abnormality or damage
- Radiotherapy, especially to the chest
- Family history of breast cancer (especially with a BRCA2 mutation)
- Klinefelter's Syndrome
Treatment
Surgery
Simple or modified radical mastectomy and surgical assessment of the lymph nodes are used when cancer is found in its early stages. Assessment of the lymph nodes is accomplished using either axillary dissection or sentinel lymph node biopsy.41, 38
Radiotherapy
Males are more likely to receive radiotherapy than females, probably due to the fact that males present with breast cancer at a later stage than females. Radiation dosages used to treat men are generally the standard amounts used to treat women. Radiotherapy has been shown effective in preventing local recurrence in men with breast cancer.41, 42
Systemic Therapy
Because about 90% of men with breast cancer are hormone receptor positive (HR+), treatment with tamoxifen is standard in HR+ men. Men treated with tamoxifen have shown higher overall survival rates. Men may also receive systemic chemotherapy.41, 39
Quality of Life
Men with breast cancer face a different environment than women coping with breast cancer. There is a large amount of information and public support for women. On the other hand there is a lack of information and added stressors for men. Men deal with issues of masculinity and stigmatism in addition to the other physical and emotional concerns that come along with having breast cancer.43, 44
Learn more about Quality of Life from CancerQuest
Resources for Men with Breast Cancer
The American Cancer Society
The National Cancer Institute
Breastcancer.org
Triple Negative Breast Cancer
Introduction
When a breast mass is diagnosed as cancerous, a sample (biopsy) of the tumor is taken and a pathologist examines its characteristics. The pathologist will determine the status of three breast cancer specific markers, the estrogen receptor (ER), progesterone receptor (PR), and a form of the epidermal growth factor receptor (HER2). These are important proteins in the design of treatment plans for breast cancers.
Triple negative (TN) breast cancer is an aggressive subtype of breast cancer that accounts for 10-15% of breast cancer cases.45, 46, 47The term "triple negative" describes tumors that do not produce significant amounts of any of the proteins listed above; TN tumors are ER minus (ER-), PR minus (PR-), and HER2 minus (HER2-). In the U.S. TN breast cancer occurs more often (>10%) in African American women than in non-African American women45, 48. The reasons for the differences in TN breast cancer occurrence in different populations is not yet clear but is an active area of research.
Karen Neely is a triple negative breast cancer survivor who was diagnosed at 34. Watch the full interview with Karen Neely.
Receptor Status
Estrogen Receptor (ER) and Progesterone Receptor (PR)
ER and PR are proteins that bind the female sex hormones estrogen and progesterone, respectively. These hormones are produced by the ovaries and play a major role in stimulating cell division in breast cells. Estrogen and progesterone bind to their respective receptors and directly stimulate genes that regulate cell division. Breast tumor cells with a positive (+) hormone receptor status have high levels of ER and PR, possibly resulting in a faster growing tumor. These types of breast cancer can be treated with hormone therapy. Triple negative breast tumors do not have high levels of these hormone receptors. Learn more about hormonal cancer treatments.
Human Epidermal Growth Factor Receptor 2 (HER2)
HER2 is a receptor protein located on the surface of breast cells. This protein binds growth factors and stimulates cell growth and division. Breast tumor cells with a positive (+) HER2 status have high levels of HER2 on their surface. This may result in an increased ability to grow and spread. These types of breast cancer can be treated with a type of targeted therapy. Triple negative breast tumors have low levels of HER2.
Prognosis
In theory triple negative tumors should have a better prognosis than tumors expressing ER, PR, or HER2, because they are not receiving the growth signals provided by these proteins, but this is not the case. Breast cancers expressing ER, PR, or HER2 can be treated with drugs that inhibit the function of the receptors (i.e. Herceptin®, tamoxifen). The triple negative subtype of breast cancer is unresponsive to the available targeted treatments and currently no specific treatment guideline exists for this tumor type45. Studies have shown that triple negative tumor cells may be more aggressive than other breast cancer subtypes, but the reasons for this are unknown 49. The lack of treatment options and aggressive nature of the tumor cells make triple negative breast cancer more difficult to treat.
Characteristics of triple negative breast cancer cells49, 50, 47:
Triple negative breast cancers tend to share additional features that can impact tumor growth or treatment. Some of these are described below:
- Nuclear Grade
The shape and size of the nucleus of a cancer cell provides an indication of how abnormal the cell is likely to behave. TN tumors are more likely to be 'high grade', indicating more severe abnormalities. - Mitotic Index
This is a measure of how rapidly the cells in the tumor are dividing. It is determined by calculating the ratio of cells dividing to cells not dividing (in the viewed samples).
A higher mitotic index may indicate a more rapidly growing tumor. TN tumors often have a high mitotic index. - Differentiation State
This describes whether or not the cancer cells 'look' like normal cells from the tissue of origin. As an example, Liver cells have specific functions and therefore do not look like breast cells. Cancer cells often lack the structure and function of the normal cells from which they arise. TN cancer cells are often 'poorly differentiated' which means they no longer look/function like normal breast cells.
Characteristics of triple negative breast cancers47, 50, 49:
- Age at Diagnosis
TN patients are often diagnosed at a slightly younger age than other breast cancer patients (average age at diagnosis: 53 vs 57.7 yrs) - Tumor Size
TN tumors tend to be larger when the cancer is detected than other breast cancer
(3.0 cm vs 2.1cm) - Tumor Grade
TN tumors more likely to be grade III than other breast cancer subtypes (66% vs 28%) - Node Positivity (regional metastasis)
lymph nodes near the tumor are more likely to test positive for the spread of cancer in TN breast cancer patients than other subtypes (54.6% vs 45.6%)
More Information:
Learn more about Herceptin®.
Learn more about Tykerb®.
Learn more about ER/PR.
Learn more about HER-2.
Breast Cancer Resources
Risks for Breast Cancer
Risk Factors Table (Susan G. Komen Breast Cancer Foundation)
Preventive Mastectomy Q&A (NCI)
Oral Contraceptives and Cancer Risk Q&A (NCI)
Detection and Diagnosis of Breast Cancer
Understanding Breast Changes (NCI)
Genetic Testing for BRCA1 & BRCA2 Mutations (NCI)
Breast Cancer Diagnosis (NBCF)
Inflammatory Breast Cancer (NCI)
Paget Disease of the Nipple Q&A (NCI)
Do It for Yourself Do It for your Family
Breast Cancer Treatments
The Study of Tamoxifen and Raloxifene (NCI)
Breast Cancer Treatment Guidelines (NCCN)
Breast Cancer Treatment and Pregnancy
Surgery Choices for Early Stage (NCI)
Breast Cancer Survivorship
Long-term Care Guidelines (ACS)
Long Term Risks for Breast Cancer Survivors
Pregnancy After Breast Cancer (ACS)
International Breast Cancer Resources
The Breast Cancer Society of Taiwan
The Breast Cancer Society of Canada
Section Summary
Introduction
- Second most prevalent cause of cancer death in women
- Most breast cancers originate in mammary ducts
Types of Breast Cancer
- In situ breast carcinoma is noninvasive. There are two types: ductal and lobular.
- Infiltrating carcinoma is invasive carcinoma. It can originate in the ducts or lobules of the breast, but has spread to the surrounding connective tissue.
Risk Factors
- A history of breast cancer greatly increases the risk for future development of breast cancer.
- It is possible to inherit defective genes that lead to the familial form of breast cancer.
- Breast cancer risk increases with age.
- African Americans women have a lower risk of developing breast cancer compared to white American women.
- Hormone replacement therapy has been associated with increased breast cancer risk, but its impact is still under investigation.
- Exposure to radiation and dietary factors also affect breast cancer risk.
Pathology Report and Staging
- A sample of breast tissue is examined micro- and macroscopically in order to create a pathology report.
- The T/N/M system is one of the most common methods used for breast cancer staging.
- The T/N/M system assigns a degree of severity based on size, lymph node involvement, and spread of the cancer.
Breast Cancer Tumor Biology
- Many genetic changes occur in cancer. Details can be found in the Mutation section.
- BRCA1, BRCA2, HER-2, PTEN, TP53, and ATM are all genes involved in breast cancer development.
- The estrogen receptor protein binds estrogen in the cell. This complex promotes cell division. The presence or absence of this protein helps determine treatment option for the cancer.
Treatment
- Treatments differ by the stage of the cancer.
- Early stage treatment involves cancers confined to the breast.
- Late stage treatment involves tumors that have spread beyond the breast.
- Treatments can include: surgery, radiation therapy, chemotherapy, hormone therapy, and targeted treatments.
Breast Cancer in Men
- Male breast cancer is rare and usually occurs later in men than women.
- The process of detection, diagnosis, and treatment is similar to those of female breast cancer.
Triple Negative Breast Cancer
- The cancer does not produce significant amounts of estrogen receptor, progesterone receptor, or a form of the epidermal growth factor receptor (HER2).
- Triple negative breast cancer tends to be an aggressive cancer.
- Triple negative breast cancer is unresponsive to the available targeted treatments.
- 1 American Cancer Society.Cancer Facts & Figures 2023. Atlanta: American Cancer Society. (2023). Retrieved from https://www.cancer.org/research/cancer-facts-statistics/all-cancer-facts-figures/2023-cancer-facts-figures.html
- 2 What is Breast Cancer? American Cancer Society (accessed July 2018). [http://www.cancer.org/docroot/CRI/content/CRI_2_4_1X_What_is_breast_cancer_5.asp]
- 3 Singh, K., Paquette, C., Kalife, E., Wang, Y., Mangray, S., Quddus, M., Steinhoff M. "Evaluating agreement, histological features and relevance of separating pleomorphic and florid lobular carcinoma-in-situ subtypes" Hum Pathol. (2018). 78:163-170. [PUBMED]
- 4ab Simpson PT, Gale T, Fulford LG, Reis-Filho JS, Lakhani SR. "The diagnosis and management of pre-invasive breast disease: pathology of atypical lobular hyperplasia and lobular carcinoma <i>in situ</i>." Breast Cancer Research (July 2003). 5(5):258-62. [PUBMED]
- 5 Breast Cancer. American Cancer Society. [http://www.cancer.org/acs/groups/cid/documents/webcontent/003090-pdf.pdf]
- 6 Barrio, A. and Zee, K. [PUBMED]
- 7 Silverstein MJ, Lagios MD. Should all patients undergoing breast conserving therapy for DCIS receive radiation therapy? No. One size does not fit all: an argument against the routine use of radiation therapy for all patients with ductal carcinoma in situ of the breast who elect breast conservation. J Surg Oncol. 2007 Jun 15;95(8):605-9. [PUBMED]
- 8 Buchholz TA, Haffty BG, Harris JR. Should all patients undergoing breast conserving therapy for DCIS receive radiation therapy? Yes. Radiation therapy, an important component of breast conserving treatment for patients with ductal carcinoma in situ of the breast. J Surg Oncol. 2007 Jun 15;95(8):610-3. [PUBMED]
- 9 Gina Kolata. Decades of Data Fail to Resolve Debate on Treating Tiny Breast Lesions. The New York Times. [http://www.nytimes.com/2015/08/22/health/study-fuels-debate-over-treating-breast-lesion-called-stage-0-cancer.html]
- 10 Wiechmann L, Kuerer HM. "The molecular journey from ductal carcinoma in situ to invasive breast cancer." Cancer. 2008 May 15;112(10):2130-42. [PUBMED]
- 11abcdef Singletary SE. "Rating the risk factors for breast cancer." Ann Surg (2003); 237(4):474-82. [PUBMED]
- 12 Newman LA, Griffith KA, Jatoi I, Simon MS, Crowe JP, Colditz GA. "Meta-analysis of survival in African American and white American patients with breast cancer: ethnicity compared with socioeconomic status." Journal of Clinical Oncology (2006); 24:1342-1349 [PUBMED]
- 13ab Whitworth A. "New research suggests access, genetic differences play role in high minority cancer death rate." Journal of the National Cancer Institute (2006); 98(10):669 [PUBMED]
- 14 Lambe M, Trichopoulos D, Hsieh CC, Wuu J, Adami HO, Wide L. "Ethnic differences in breast cancer risk: a possible role for pregnancy levels of alpha-fetoprotein?" Epidemiology (2003);14(1):85-9. [PUBMED]
- 15 Rao CV, Li X, Manna SK, Lei AM, Aggarwal BB. "Human Chorionic Gonadotropin Decreases Proliferation and Invasion of Breast Cancer MCF-7 Cells by Inhibiting NF-kB and AP-1 Activation." J Biol Chem (2004), Vol. 279, Issue 24, 25503-25510. [PUBMED]
- 16 Rosenberg LU, Magnusson C, Lindstrom E, Wedren S, Hall P, Dickman PW. "Menopausal hormone therapy and other breast cancer risk factors in relation to the risk of different histological subtypes of breast cancer: a case-control study. Breast Cancer Research (2006); 8(1) R11 [PUBMED]
- 17 Veurink M, Koster M, Berg LT. "The history of DES, lessons to be learned." Pharm World Sci (2005) 27:139-143. [PUBMED]
- 18 Travis LB, Hill D, Dores G, Gospodarowicz M, Leeuwen F, Holowaty E, Glimelius B Andersson M, Pukkala E, Lynch C, Pee D, Smith S, Veer M, Joensuu T, Storm H, Stovall M, Boice J, Gilbert E, Gail M. "Cumulative absolute breast cancer risk for young women treated for Hodgkin lymphoma." J of National Cancer Institute (2005) 97(19):1428-1435 [PUBMED]
- 19 Felton JS, Knize MG, Salmon CP, Malfatti MA, Kulp KS. "Human Exposure to Heterocyclic Amine Food Mutagens/ Carcinogens: Relevance to Breast Cancer." Environmental and Molecular Mutagenesis (2002) 39:112-118 [PUBMED]
- 20 https://www.sciencedaily.com/releases/2017/05/170523084758.htm
- 21 American Cancer Society. Cancer Facts & Figures 2010. Atlanta: American Cancer Society; 2010. [http://www.cancerquest.org/sites/default/files/assets/pdf/facts%26figures2010.pdf]
- 22 Cancer Facts and Figures 2013. American Cancer Society. [http://www.cancer.org/acs/groups/content/@epidemiologysurveilance/documents/document/acspc-036845.pdf]
- 23 "Mammography and Other Breast Imaging Procedures." American Cancer Society (6-19-2002). [http://www.cancer.org/Healthy/FindCancerEarly/ExamandTestDescriptions/MammogramsandOtherBreastImagingProcedures/mammograms-and-other-breast-imaging-procedures]
- 24 Blamey RW, Wilson ARM, Patnick J. "Screening for Breast Cancer." British Medical Journal (September 2000). 321(7262): 689-693. [PUBMED]
- 25 Mettler FA, Upton AC, Kelsey CA, Ashby RN, Rosenburg RD, Linver MN. "Benefits Versus Risks from Mammography: A Critical Reassessment." Cancer (1996). 77(5): 903-909. [PUBMED]
- 26 Saslow D, Boetes C, Burke W, Harms S, Leach MO, Lehman CD, Morris E, Pisano E, Schnall M, Sener S, Smith RA, Warner E, Yaffe M, Andrews KS, Russell CA; American Cancer Society Breast Cancer Advisory Group. American Cancer Society guidelines for breast screening with MRI as an adjunct to mammography. CA Cancer J Clin. 2007 May-Jun;57(3):185. [PUBMED]
- 27 Ross JS, Fletcher JA, Linette GP, Stec J, Clark E, Ayers M, Symmans WF, Pusztai L, Bloom KJ. "The HER-2/neu gene and protein in breast cancer 2003: biomarker and target of therapy." The Oncologist (2003); 8:307-325 [PUBMED]
- 28 Sabate JM, Gomez A, Torrubia S, Blancas C, Sanchez G, Alonso MC, Lerma E. "Evaluation of breast involvement in relation to Cowden syndrome; a radiological and clinicopathological study of patients with PTEN germ-line mutations." Eur Radiol (2006) 16:702-706 [PUBMED]
- 29abc Thull DL, Vogel VG. "Recognition and Management of Hereditary Breast Cancer Syndromes." The Oncologist (2004) 9:13-24 [PUBMED]
- 30 Cuatrecasas M, Santamaria G, Velasco M, Camacho E, Hernandez L, Sanchez M, Orrit C, Murcia C, Cardesa A, Campo E, Fernandez PL. "ATM gene expression is associated with differentiation and angiogenesis in ingiltrating breast carcinomas." Histology and Histopathology (2006) 21:149-156 [PUBMED]
- 31ab Mackay J, Taylor A. "Moving genetics into clinical cancer care: examples from BRCA gene testing and telemedicine." Breast. 2006 Dec;15 Suppl 2:S65-70. [PUBMED]
- 32abc Marshall M, Solomon S. "Abstract Hereditary breast-ovarian cancer: clinical findings and medical management." Plast Surg Nurs. 2007 Jul-Sep;27(3):124-7. Review. [PUBMED]
- 33 Arpino G, Wiechmann L, Osborne CK, Schiff R. "Crosstalk between the estrogen receptor and the HER tyrosine kinase receptor family: molecular mechanism and clinical implications for endocrine therapy resistance." Endocr Rev. 2008 Apr;29(2):217-33. [PUBMED]
- 34 Skliris GP, Leygue E, Watson PH, Murphy LC. "Estrogen receptor alpha negative breast cancer patients: estrogen receptor beta as a therapeutic target." Am J Physiol Endocrinol Metab. 2008 Jun 24. [PUBMED]
- 35 Isern AE, Loman N, Malina J, Olsson H, Ringberg A. "Histopathological findings and follow-up after prophylactic mastectomy and immediate breast reconstruction in 100 women from families with hereditary breast cancer." Eur J Surg Oncol. 2008 Apr 21. [PUBMED]
- 36 Poggi MM, Danforth DN, Sciuto LC, Smith SL, Steinberg SM, Liewehr DJ, Menard C, Lippman ME, Lichter AS, Altemus RM. "Eighteen-Year Results in the Treatment of Early Breast Carcinoma with Mastectomy versus Breast Conservation Therapy:the National Cancer Institute Randomized Trial." Cancer. 2003 Aug 15;98(4):697-702. [PUBMED]
- 37 McCready D, Holloway C, Shelley W, Down N, Robinson P, Sinclair S, Mirsky D. "Surgical management of early stage invasive breast cancer: a practice guideline." Can J Surg. 2005 Jun;48(3):185-94. [PUBMED]
- 38abcdefg IS Fentiman, A Fourquet, GN Hortobagyi. Male Breast Cancer. Lancet. 2006; 367(9510): 595-604. [PUBMED]
- 39abcdef SH Giordano, AU Buzdar, GN Hortobagyi. Breast Cancer in Men. Annals of Internal Medicine. 2002; 137: 678-687. [PUBMED]
- 40 American Cancer Society. Cancer Facts & Figures 2016. Atlanta: American Cancer Society; 2016. [http://www.cancer.org/acs/groups/content/@research/documents/document/acspc-047079.pdf]
- 41abc Agrawal A, AA Ayantunde, R Rampaul, JF Robertson. Male Breast Cancer: A Review of Clinical Management. Breast Cancer Research and Treatment. 2007; 103: 11-21. [PUBMED]
- 42 G Macdonald, C Paltiel, IA Olivotto, S Tyldesley. A Comparative Analysis of Radiotherapy Use and Patient Outcome in Males and Females with Breast Cancer. Annals of Oncology. 2005; 16:14421448. [PUBMED]
- 43 Brian K, Williams B, Iredale R, France L, Gray J. Psychological Distress in Men With Breast Cancer. 2006; 24: 95-101. [PUBMED]
- 44 France L, Michie S, Barrett-Lee P, Brain K, Harper P, Gray J. Male Cancer: A Qualitative Study of Male Breast Cancer. 2000; 9: 343-348. [PUBMED]
- 45abc Cleator S, Heller W, Coombes RC. Triple-negative breast cancer: therapeutic options. Lancet. 2007; 8: 235-44. [PUBMED]
- 46 Sasa M, Bando Y, Takahashi M, Hirose T, Nagao T. Screening for basal marker expression is necessary for decision of therapeutic strategy for triple-negative breast cancer. 2007 [E-pub ahead of print]. [PUBMED]
- 47abc Dent R, Trudeau M, Pritchard KI, Hanna WM, Kahn HK, Sawka CA, Lickley LA, Rawlinson E, Sun P, Narod SA. Triple-negative breast cancer: clinical features and patterns of recurrence. Clinical Cancer Research. 2007; 13(15 Pt1): 4429-4434. [PUBMED]
- 48 Demicheli R, Retsky MW, Hrushesky WJ, Baum M, Gukas ID, Jatoi I. Racial disparities in breast cancer outcome: insights into host-tumor interactions. Cancer. 2007; 110(9): 1880-1888. [PUBMED]
- 49abc Carey LA, Perou CM, Livasy CA, Dressler LG, Cowan D, Conway K, Karaca G, Troester MA, Tse CK, Edmiston S, Deming SL, Geradts J, Cheang MC, Nielsen TO, Moorman PG, Earp HS, Millikan RC. Race, breast cancer subtypes, and survival in the Carolina Breast Cancer Study. JAMA. 2006; 295(21):2492-502. [PUBMED]
- 50ab Bauer KR, Brown M, Cress RD, Parise CA, Caggiano V. Descriptive analysis of Estrogen Receptor (ER)-Negative, Progesterone Receptor (PR)-Negative, and HER2-Negative Invasive Breast Cancer, the so-called Triple-Negative Phenotype. Cancer. 2007; 109:1721-1728. [PUBMED]
