Cervical cancer forms in the interior lining of the cervix, the junction of the vagina and uterus. The development of cervical cancer is typically slow, and occurs over a period of years. The progression to cervical cancer begins with the development of precancerous changes in normal cells. Most of these changes, even if left untreated, will not progress to cancer. However, in 2023 the American Cancer Society estimates 13,960 women will be diagnosed with invasive cervical cancer and 4,310 will die of the disease in the United States.1

Cervical cancer is most often diagnosed in middle-aged women, with half of those diagnosed between the ages of 35 and 55. Cervical cancer is very rarely seen in women less than 20 years of age, but approximately 20% of cases occur in women over 65, demonstrating the necessity of continued screening procedures. The five year survival rate of invasive cervical cancer is currently 67% and improvements in screening and the development of prophylactic vaccines have decreased the incidence of late-stage cancer.2
The following sections can be found on this page:
Interactive Games: Teaching Materials: |
Watch an interview with cervical cancer specialist Dr. Ira Horowitz.
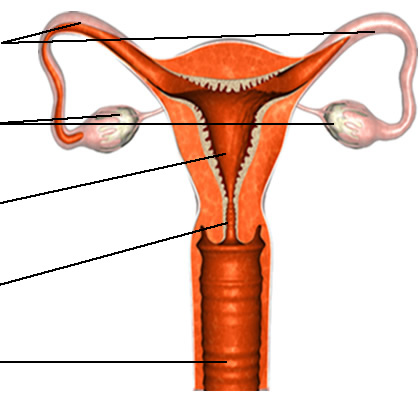
Anatomy Of The Female Reproductive System
The cervix is part of the female reproductive tract that makes up the lower part of the uterus, also known as the womb. The upper endocervix connects to the uterus and the lower ectocervical region opens to the vagina. This connection allows for the passage of a fetus during delivery.3
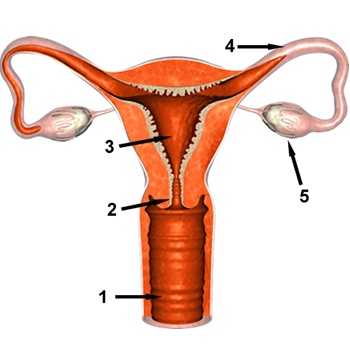
The graphic above depicts the major components of the female reproductive tract.
- Vagina - female sex organ, passageway for menstrual blood and fetus during childbirth
- Cervix - region connecting the uterus to the vagina; the muscles of the cervix support the weight of the fetus during pregnancy
- Uterus (womb) - location where fertilized egg develops into a fetus and is nourished until birth. Note that the walls of the uterus are thick and lined with muscles
- Fallopian (uterine) Tube - location of fertilization of an egg by a sperm cell
- Ovary - produce and store gametes (eggs) and produce the female sex hormones, estrogen and progesterone
Types Of
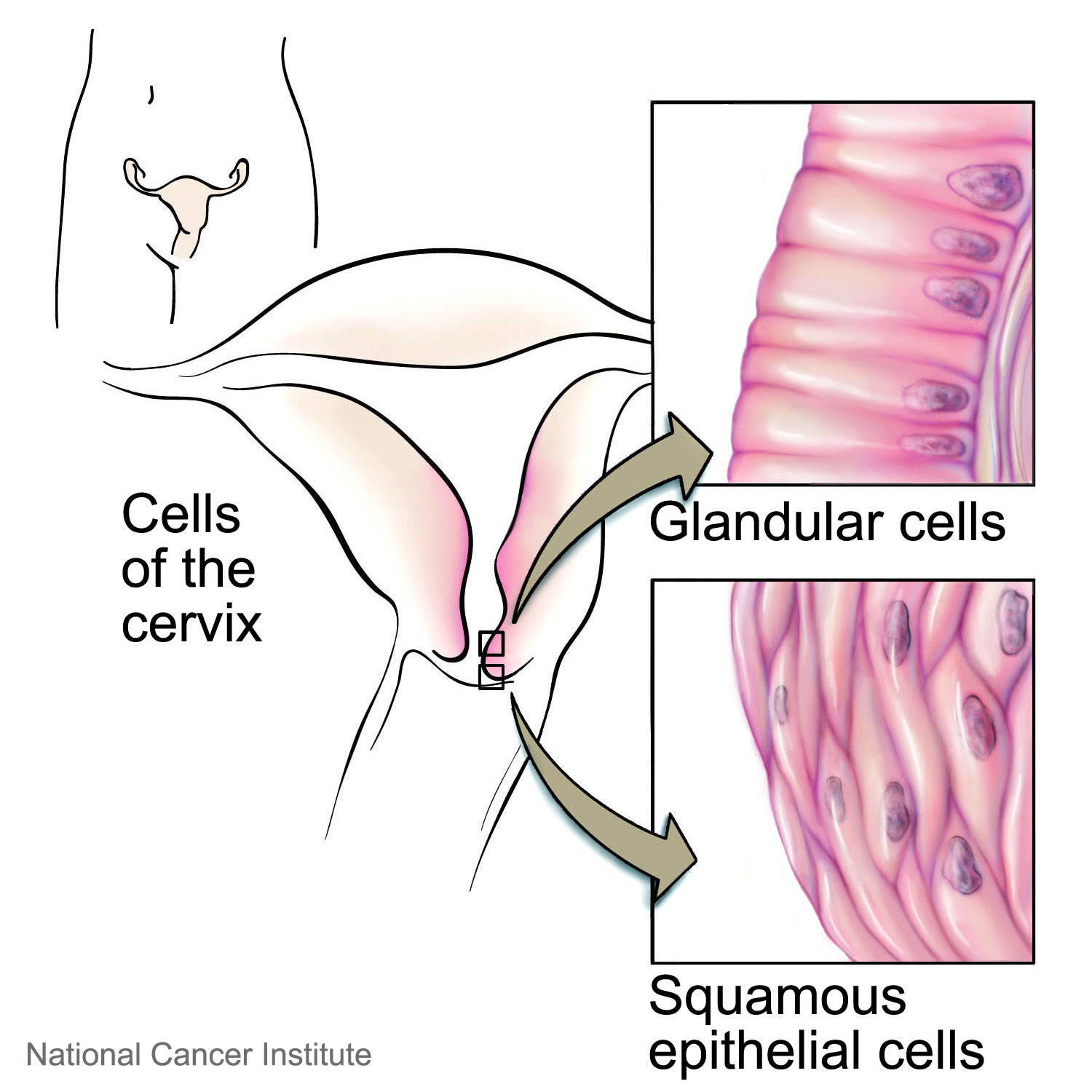
The two types of cervical cancer are squamous cell carcinoma and adenocarcinoma, which are distinguished based on their appearance under a microscope. Both squamous cell and adenocarcinoma begin in the cells that line hollow organs, but squamous cells have a thin, flat appearance while adenocarcinomas involve cells with secretory functions. Squamous cell carcinoma is far more common and makes up approximately 90% of cervical carcinoma cases. Both types have similar risk factors, prognoses and treatments.4
Hear how cervical cancer survivor Lori Grice reacted to her diagnosis of adenocarcinoma by clicking on the image below. You can also watch the full interview with Lori and DeWayne Grice.
Risk Factors
There are several risk factors for the development of cervical cancer, both genetic and environmental. These include:
- Human Papillomavirus (HPV) Infection
- Family History of Cervical Cancer
- Age
- Sexual and Reproductive History
- Socioeconomic Status
- Smoking
- HIV Infection
- In Utero DES Exposure
- Long-term use of oral contraceptives
Human Papillomavirus (HPV) Infection:

HPV infection is associated with virtually all cases of cervical cancer. HPV is among the most common sexually transmitted diseases and most women clear the infection within two years without complications. Long term infection with high-risk strains of HPV can lead to the development of cervical dysplasia and cancer.5
Because of the very high correlation between HPV infection and cervical cancer, the following paragraphs describe HPV in more detail.
The Human papillomaviruses are a family of sexually transmitted viruses consisting of over 100 different viral strains, 40 of which are known to infect the human genital tract and 15 of which have been associated with cervical cancer. Most patients with one of these infections show no symptoms, but some strains of HPV lead to the development of genital warts.
HPV contains a small, circular, double stranded DNA genome. The virus infects epithelial cells, one of the rapidly dividing cells that form the skin and mucous membranes. The virus reproduces within the host cell and when the cell dies, as part of natural cell turnover, the new virus particles (virions) are released and can infect other cells. The DNA of low-risk types of HPV remains separate from the host DNA, while high-risk forms are able to combine with (insert into) the host DNA. Insertion into the host genome is problematic because it interrupts the regulation of the viral genes. Without this control, the viral genome is transcribed at a much higher rate.
The HPV genome contains at least two genes whose protein products function as oncogenes, meaning that they can turn into cancer cells. These genes are called E6 and E7. The E6 and E7 proteins inhibit the tumor suppressor proteins p53 and pRb, respectively. By 'turning off' the p53 gene, cells will not die (this process is called immortalization). The inactivation of pRb leads to increased cell division. While either one of these mutations has the potential to lead to the development of cancer, the ability of HPV to inactivate both tumor suppressors further increases the efficiency of the transformation from normal to cancerous cells.6
Infection with low-risk types of HPV generally produce benign lesions with a minimal chance of progression to dysplasia or cancer. However, high risk HPV strains (16, 18, 31, 33 and 35) are implicated in 99% of those diagnosed with cervical cancer. It is important to note that most women infected with high-risk strains of HPV infection will not develop cancer. The risk of developing dysplasia or cancer after HPV infection is, in part, dependent on the amount of virus present during infection and the length of time it takes to clear the infection.6
There is no cure or treatment for HPV infection. Even without treatment, most infections are cleared by the immune system within two years. If the infection persists there is an increased chance of viral DNA integration and progression to cancer. 5Women can be tested to learn if they are infected with HPV. Even though there is currently no cure for HPV infection, the knowledge can help women make responsible choices regarding their sexual practices. Furthermore, it is possible to prevent HPV infection by receiving a vaccination. Vaccinations are recommended for young individuals—both female and male--within their early teens or late 20’s.7
More on Tumor Suppressors and Oncogenes
Family History of Cervical Cancer:
Women with a family history of cervical cancer, especially an affected mother or sister, have a two-fold risk of developing cervical cancer, suggesting an inherited susceptibility. However, there does not appear to be a correlation between a family history of other cancer types (i.e. colon cancer) and the risk of developing cervical cancer.8
Age:
Very few women under the age of 20 are diagnosed with cervical cancer and more than half of those diagnosed are between the ages of 35 and 55. The risk decreases after age 55, but 20% of cases occur in women over 60 years old. The pattern seen is due to two conflicting factors, 1) changes in sexual behaviors and 2) the tendency of genetic mutations to accumulate over time.9
Watch the full interview with Dr. Ira Horowitz.
Sexual and Reproductive History:
Epidemiological studies have shown an increased risk for invasive cervical cancer attributable to sexual and reproductive behavior. Increased numbers of sexual partners and lower age at first sexual act have both been associated with increased risk. Women who have had multiple pregnancies and are younger at the time of their first full-term pregnancy also demonstrate an increased risk. Long term use of oral contraceptives has been shown to increase risk in some studies, but this remains controversial. More recent studies have suggested that women who have used oral contraceptives for 5 or more years have a higher risk of cervical cancer than women who have never used contraceptives. While cervical cancer risk appears to increase the longer a woman uses contraceptives, the risk appears to gradually decline after use of oral contraceptives ceases.10, 11
Because HPV is a sexually transmitted disease, behaviors that increase sexual contacts are considered risk factors.12
Socioeconomic Status:
Low socioeconomic status has proven to be a significant risk factor for invasive cervical cancer due to its large impact on education and medical resources. Results of the analysis of several epidemiological studies indicate that Hispanic and African-American women have a higher risk of invasive cervical cancer than Caucasian women.13
Decreased risk is associated with increased education--women without a college degree have an increased risk, regardless of race. Therefore, it is possible that if access to screening and medical education were equalized, race would not prove to be a significant risk factor. The increased risk with low socioeconomic status is attributed to a lack of screening, failure to treat precancerous conditions, and lack of knowledge about prevention of HPV infection.14, 5
Smoking:
Current smoking is a risk factor for the development cervical cancer due to the ability of carcinogens in cigarette smoke to cause mutations in DNA. This can also result in suppression of the immune system, which can allow HPV infections to persist and even progress into lesions or invasive cancer. In the epidemiological studies that have been conducted, smoking was associated with an increased risk of squamous cell carcinoma of the cervix, but not adenocarcinoma. Secondhand smoking also appears to increase risk of cervical cancer, even among non-smokers. A study examining the effects of secondhand smoking on cervical cancer risk found that non-smokers exposed to at least 2 hours of secondhand smoking per day tended to be at greater risk for CIN 1 cervical cancer.5, 15
Human Immunodeficiency Virus (HIV):
Women infected with HIV have been shown to have a five-fold risk of developing cervical cancer. HIV weakens the immune system, decreasing the ability to fight infection; therefore HPV infections are more likely to persist. This is thought to provide more time for the HPV to induce cancer. The high correlation between HIV infection and HPV infection is also partly due to the fact that both are sexually transmitted diseases and behaviors that put women at risk for one also put them at risk for the other.16, 17
In Utero Diethylstilbestrol (DES) Exposure:
DES is a synthetic estrogen used from the 1930s to the 1970s to reduce complications during pregnancy. Use of this drug was discontinued after it was demonstrated that the drug could harm the developing baby. Elevated risk of cervical cancer is just one of the potential health effects for women who where exposed to DES while they were in their mothers womb; others include a variety of gynecological cancers, reproductive tract irregularities, infertility and complications during pregnancy.18
Prevention
As described previously, the major risk factor for developing cervical cancer is infection with the human papillomavirus (HPV).
Abstinence is the only sure way to prevent infection with the virus. For sexually active women, the use of a condom can reduce, but not eliminate, risk of infection.
Two vaccines are available that prevent infection with the forms of the virus most associated with cervical cancer.
Visit the cancer prevention section to learn more about cervical cancer vaccines.
Watch the full interview with Dr. Ira Horowitz
In 2017, a study of 12,000 women demonstrated a link between the use of intrauterine devices (IUDs) and a reduction in cervical cancer rates. Women using the device had a one-third reduction (33%) in their rate of cervical cancer. The study was not designed to show that IUD use caused the reduction, but the link was identified. Researchers suggest that the presence of the IUD in the uterus causes changes in the immune system that help users fight off HPV infection.19
Symptoms
Symptoms:
Typically, early cervical cancer is asymptomatic (no symptoms), but abnormal vaginal bleeding can occur once the cancer becomes invasive.20, 9
Watch the video to learn about HPV infection symptoms from cervical cancer specialist Dr. Kevin Ault.
Click here to watch the full interview with Dr. Ault.
Detection And Diagnosis
Detection:
Since early cervical cancer is asymptomatic, regular screening by a Pap smear is important in order to avoid the progression of precancerous lesions or noninvasive cancer. Current US guidelines, released March 2012, recommend Pap smears for women over 21. Screening should then be repeated every three years until at least age 65. If you have had a history of normal Pap smears and a radical hysterectomy, in which the cervix has been removed due to non-cancerous reasons (i.e., fibroids), routine screening via Pap smear may not be necessary. The American Cancer Society recommends screening with Pap smear and HPV DNA testing every five years from age 30-65 but finds a Pap smear every three years acceptable. Women with prenatal exposure to diethylstilbestrol (DES), a prior history of cervical cancer, or a disease/condition that weakens their immune system, should continue screening past age 65. Although cancers of the ovaries, cervix, and uterus may be difficult to detect at an early stage, there are several detection options currently in use.21
There is some evidence that testing for HPV may be a better way to prevent the development of cancer than Pap smears22, but these results need to be confirmed before any recommendations are changed.
Important tests for detecting cancers of the female reproductive tract include:
- Pap Smear for detecting cervical and vaginal cancer.
- Ultrasound for detecting ovarian and uterine cancer.
- CA-125 for detecting ovarian cancer.
If the diagnostic tests indicate the presence of cancer, additional imaging (CT, MRI, etc.) may be performed to determine the location and extent of the disease.20, 23, 24
Specific tests for Human Papillomavirus (HPV) DNA may be administered annually to women over 30, which lessens the necessity of Pap smears to once every five years. 25, 9, 26HPV screening involves a Polymerase Chain Reaction (PCR) based test that is able to detect DNA sequences common to all HPV strains. Because this sequence can be disrupted when the virus inserts itself into cellular DNA, other genes (E6 and E7) are often also identified. This allows the tests to reliably detect the presence of high-risk HPV strains. In a recent study, the addition of HPV DNA tests to traditional Pap smears reduced the incidence of grade 2/3 cervical intraepithelial neoplasia (CIN) by 40%. 27Since HPV infections are frequently transient, it is possible that risk for cervical dysplasia or cancer may exist even with negative test results.6
More on Viruses and Cancer
More on the Pap smear
Diagnosis:
If a Pap smear reveals abnormal cells, further diagnostic tests are performed to determine a diagnosis. Irregular cells could indicate:
- Human Papillomavirus Infection
- Cervical Intraepithelial Neoplasia (CIN)
- Cervical Cancer
Watch the full interview with cervical cancer advocates Lori and DeWayne Grice.
Further tests are necessary to make a diagnosis. Additional tests that may be performed include:
- Colposcopy - a camera is inserted into the vagina and the cervix is examined.
- Biopsy - removal of a small sample of tissue for examination by a pathologist. The procedure is typically done without anesthesia and is associated with minimal pain or bleeding. It is possible to perform the biopsy during a colposcopy.
More on General Detection Techniques.
Pathology Report And Staging
Cervical Intraepithelial Neoplasia:
Cervical intraepithelial neoplasia (CIN) is an abnormal condition that is detectable by Pap smears and other cervical exams. CIN is the growth of abnormal cells in the lining of the cervix. Though CIN it is not cancerous, it has the potential to progress to cancer if left untreated. There are three stages (or grades) of CIN: CIN 1, CIN 2 and CIN 3. The stages are define by how abnormal the cells appear, slight, moderate and high. The risk of cancer development increases with increasing CIN grade. CIN is relatively common, with 1.4 million low grade and 330,000 high grade cases diagnosed in the United States in 2006. Cervical lesions are treated depending on the degree of severity. CIN 1 lesions may be removed or closely monitored; CIN 2/3 lesions are usually surgically removed. In either case, careful follow-up screening is performed to ensure that there is no recurrence. Despite the high incidence of CIN, if these irregularities are treated, progression to cancer is very rare. 28The image below shows microscopic images of normal cervical tissue, CIN 1, CIN 2 and CIN 3
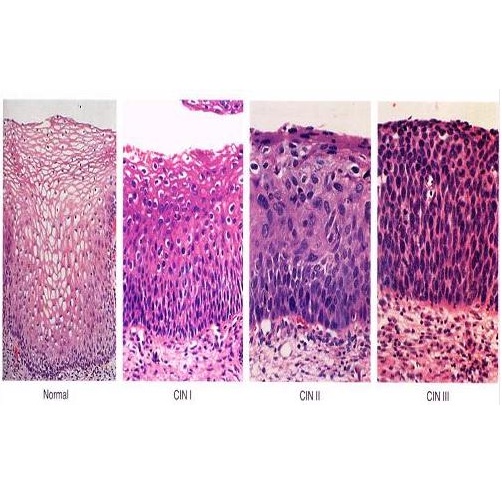
Image courtesy of Talaat S Tadros MD, Emory University School of Medicine.
Cervical Cancer Staging:
If a lesion is determined to be cancerous, the disease is staged. Staging is typically based on guidelines produced by the Federation Internationale de Gynecologie et d' Obstetrique (FIGO). In this system tumors are classified by their size and location. An alternative system is used by the American Joint Committee on Cancer (AJCC). 29Staging of the cancer helps the clinicians to design an appropriate plan of treatment.
View the FIGO guide to cervical cancer staging.
Visit the AJCC website.
Treatment
Depending on the stage and location of the cancer, several different treatments are used for cervical cancer. Common treatment methods are listed below.20, 23, 24
Surgery:
Cryosurgery - used for pre-invasive cancer; kills cancer cells by freezing them with a metal probe that has been cooled by liquid nitrogen.
Laser Surgery - used for pre-invasive cancer; laser beams are used to burn off abnormal cells or remove tissue for further study.
Cone Biopsy - used to preserve fertility in women with early stage cancer or to obtain a sample for further study; a cone-shaped section of tissue is taken from the cervix.
Simple Hysterectomy - the uterus and cervix are removed either through the vagina or the abdominal wall; results in infertility.
Radical Hysterectomy and Pelvic Lymph Node Dissection - the uterus, upper vagina and lymph nodes are removed through the vagina or the abdominal wall; results in infertility.
Pelvic Externation - for recurrent cancer; radical hysterectomy is accompanied by removal of the bladder, vagina, rectum and part of the colon; results in infertility.For details on surgical treatment view the section on Surgery.
Radiation:
Either internal (also called brachytherapy) or external radiation may be used to treat cervical cancer. For details on these treatments view the section on Radiotherapy.
Chemotherapy:
Several different drugs may also be used to treat primary or recurrent cervical cancer. For details on specific chemotherapy treatments view the section on Chemotherapy.
NOTE: Sometimes radiation and chemotherapy are given at the same time. The rationale is that the low levels of chemotherapy given make the cancer cells more sensitive to the radiation. The technique is called chemoradiotherapy or radiosensitization.30
If a woman is pregnant at the time of diagnosis, a decision must be made, based on the stage of the cancer, whether to continue the pregnancy to term. Treatment for cervical cancer cannot be administered during pregnancy, and aggressive cancers often require immediate treatment.
Watch the entire interview with cervical cancer survivor Lori Grice and her husband DeWayne Grice.
Information about clinical trials:
- General clinical trial information from CancerQuest
- Click here for information about clinical trials from Georgia Clinical Trials Online.
- Click here for information about clinical trials from the National Cancer Institute.
The Society of Gynecologic Oncology has produced a series of educational publications on cervical cancer, uterine cancer, ovarian cancer, vaginal cancer, and vulvar cancer. Their materials include planning guides and survivorship care plans.
Cervical Cancer Resources
Risks for Cervical Cancer
National Cervical Cancer Coalition (NCCC)
Oral Contraceptives and Cancer Risk Q&A (NCI)
Detection and Diagnosis of Cervical Cancer
Winship Cancer Institute: Cervical Cancer Diagnosis and Staging Make an Appointment
Cervical Cancer Treatments
Understanding Cervical Changes
Cervical Cancer Treatment (NCI)
Cervical Cancer Survivorship
Survivorship Support For Cervical Cancer
Long Term Risks for Cervical Cancer Survivors
Radiation Therapy for Cervical Cancer
International Cervical Cancer Resources
What You Need to Know: Cervical Cancer - Vietnamese
Cervical Cancer Treatment in Taiwan
Cervical Cancer - Canadian Cancer Society
Public Health Agency of Canada: Cervical Cancer
Cervical Cancer: Cancer Council Australia
Section Summary
Introduction
- The cervix is the junction of the vagina and the uterus.
- Cervical cancer forms on the interior lining of the cervix.
- Typically, cervical cancer development is slow, occurring over a period of years.
Types of Cervical Cancer
- There are two main types of cervical cancer: squamous cell carcinoma and adenocarcinoma.
- Squamous cell carcinoma is the most common type.
Risk Factors
- Human papillomavirus (HPV) infection is associated with virtually all cases of cervical cancer.
- HPV is sexually transmitted, but only 15 of over 100 strains are implicated as causing cervical cancer.
- A family history of cervical cancer doubles the risk for developing the cancer.
- Most cases of cervical cancer occur after the age of 20.
- Increased numbers of sexual partners and lower age at first sexual act have both been associated with increased risk.
- Smoking has been associated with an increased risk of squamous cell carcinoma of the cervix.
- Women infected with HIV have been shown to have a five-fold risk of developing cervical cancer.
- The long-term use of oral contraceptives has been shown to increase the risk.
Symptoms
- Early cervical cancer is asymptomatic.
- Abnormal vaginal bleeding can occur once the cancer becomes invasive.
Detection and Diagnosis
- Cervical cancer can be detected by a Pap smear.
- HPV tests are available to detect the presence of viral DNA.
- More tests are carried out if the Pap smear reveals abnormal cells.
- A colposcopy can be used to view the tissue of the cervix.
- A tissue biopsy can be done and a pathologist can then examine the sample.
- Additional imaging (CT, MRI, etc.) may be performed.
Pathology Report and Staging
- Cervical intraepithelial neoplasia (CIN) is a non-cancerous, abnormal growth of cells lining the cervix.
- The risk of cancer development increases with increasing CIN grade.
- Cervical cancer staging is determined by the size and location of the tumor.
Treatment
- Different types of surgery can be performed depending on the stage of the cancer: cryosurgery, laser surgery, cone biopsy, simple hysterectomy, radical hysterectomy, and pelvic externation.
- Internal or external radiation and chemotherapy are possible treatments for cervical cancer.
Cervical Cancer Vaccine
- The vaccine Gardasil® has been approved in the U.S. for males and females aged 9-26 for the prevention of infection by HPV 6, 11, 16, and 18.
- The vaccine Cervarix® has been approved in the U.S. for females aged 10-25 for the prevention of infection by HPV 16 and 18.
- The vaccines prevent new infections and are not effective against pre-existing HPV infections.
Know The Flow
Know the Flow is an educational game for you to test your knowledge. To play:
- Drag the appropriate choices from the column on the right and place them in order in the boxes on the left. Note that you will only use five of the six choices to complete the game.
- When done, click on 'Check' to see how many you got correct.
- For incorrect answers, click on 'Description' to review information about the processes.
- To try again, choose 'Reset' and start over.
Please visit us on a larger screen to play this game.
Pick A Part: Female Reproductive System
Pick A Part is an educational game for you to test your knowledge. To play:
- Drag the appropriate choices from the column on the bottom and place them in order in the boxes on the left. Note that you will only use five of the six choices to complete the game.
- When done, click on 'Check' to see how many you got correct.
- For incorrect answers, click on 'Description' to review information about the processes.
- To try again, choose 'Reset' and start over.
Please visit us on a larger screen to play this game.
For help please refer to the Female Reproductive System page.
The Grid
The GRID is an educational quiz game, covering cancer related topics. The format of the game is similar to a popular TV quiz show, where the clues are phrased as an answer and the response os phrased in the form of a question. For example:
Clue: This website won the 2007 "Cancer on the Internet" award.
Response: What is CancerQuest?
How to play:
For one person:
Enter your name and begin the game. Choose a category and a point value, the clue will open. To check your response, click and hold the "Ans" button. If your response was correct click the "Check", the point value will be added to your total. If your response was incorrect click the "X", the point value will be subtracted from your total (you lose points for incorrect responses).
For two or more:
Enter player's names and begin the game (click the "make active" box to add players). Choose a category and a point value, the clue will open. Each player uses a different keyboard key to "ring in"
- Player 1 - use "z" key
- Player 2 - use "." (period) key
- Player 3 - use "enter" key on far right of keyboard
To check response click and hold the "Ans" button. If a correct response is given, click the "Check". The point value will be added to the responding player's total. If an incorrect response is given, click the "X". The point value will be subtracted from responding player's total (an incorrect response results in a loss of points). The player with the correct response chooses the next clue. Continue until all clues are uncovered, the player with the most points wins the game.
Please visit us on a larger screen to play this game.
- 1 American Cancer Society.Cancer Facts & Figures 2023. Atlanta: American Cancer Society. (2023). Retrieved from https://www.cancer.org/research/cancer-facts-statistics/all-cancer-facts-figures/2023-cancer-facts-figures.html
- 2 Cervical Cancer: Statistics. Cancer.Net. Accessed 5 September 2018. [https://www.cancer.net/cancer-types/cervical-cancer/statistics]Arbyn, M., Xu, L., Simoens, C., and Martin-Hirsch, PP. "Prophylactic vaccination against human papillomaviruses to prevent cervical cancer and its precursors." 2018. Cochrane Database Syst Rev. 9;5:CD009069. [PUBMED.]
- 3 Urry, L. A., Cain, M. L., Wasserman, S. A., Minorsky, P. V., & Reece, J. B. (2017). Campbell Biology (11th ed.). Pearson.
- 4 National Cancer Institute (Accessed 7 September 2018) [http://www.cancer.gov/cancertopics/understandingcervicalchanges/page2]
- 5abcd Khan MJ, Partridge EE, Wang SS, Schiffman M. Socioeconomic status and the risk of cervical intraepithelial neoplasia grade 3 among oncogenic human papillomavirus DNA-positive women with equivocal or mildly abnormal cytology. Cancer. 2005 Jul 1;104(1):61-70. [PUBMED]
- 6abc Boulet G, Horvath C, Broeck DV, Sahebali S, Bogers J. Human papillomavirus: E6 and E7 oncogenes. Int J Biochem Cell Biol. 2007;39(11):2006-11. Epub 2007 Jul 19. [PUBMED]
- 7 Petrosky, E., Bocchini JA Jr., Hariri, S., Chesson, H., Curtis CR., Saraiya M., Unger, ER., Markowitz, LE., Centers for Disease Control and Prevention (CDC). "Use of 9-valent human papillomavirus (HPV) vaccine: updated HPV vaccination recommendations of the advisory committee on immunization practices." 2015. MMWR Morb Mortal Wkly Rep. 64(1): 300-304. [PUBMED.]
- 8 Negri E, La Vecchia C, Bosetti C, Franceschi S, Parazzini F. Risk of cervical cancer in women with a family history of breast and female genital tract neoplasms. Int J Cancer. 2005 Dec 10;117(5):880-1. [PUBMED]
- 9abc American Cancer Society (accessed 9/5/07) [http://www.cancer.org]
- 10 ”Oral Contraceptives and Cancer Risk." Accessed 8 September 2018. National Cancer Institute. [https://www.cancer.gov/about-cancer/causes-prevention/risk/hormones/oral-contraceptives-fact-sheet.]
- 11 International Collaboration of Epidemiological Studies of Cervical Cancer, Appleby P, Beral V, Berrington de González A, Colin D, Franceschi S, Goodhill A, Green J, Peto J, Plummer M, Sweetland S. "Cervical cancer and hormonal contraceptives: collaborative reanalysis of individual data for 16,573 women with cervical cancer and 35,509 women without cervical cancer from 24 epidemiological studies." Lancet. 2007 Nov 10;370(9599):1609-21 [PUBMED]
- 12 International Collaboration of Epidemiological Studies of Cervical Cancer. Comparison of risk factors for invasive squamous cell carcinoma and adenocarcinoma of the cervix: collaborative reanalysis of individual data on 8,097 women with squamous cell carcinoma and 1,374 women with adenocarcinoma from 12 epidemiological studies. Int J Cancer. 2007 Feb 15;120(4):885-91. Erratum in: Int J Cancer. 2007 Jun 1;120(11):2525. [PUBMED]
- 13 Markowitz LE, Dunne EF, Saraiya M, Lawson HW, Chesson H, Unger ER; Centers for Disease Control and Prevention (CDC); Advisory Committee on Immunization Practices (ACIP).Quadrivalent Human Papillomavirus Vaccine: Recommendations of the Advisory Committee on Immunization Practices (ACIP). MMWR Recomm Rep. 2007 Mar 23;56(RR-2):1-24. [PUBMED]
- 14 Shin, H., Kang, H., Lee, J., Lim, H. "The Association between Socioeconomic Status and Adherence to Health Check-up in Korean Adults, Based on the 2010-2012 Korean National Health and Nutrition Examination Survey." 2018. Korean J Fam Med. 39(2): 114-121. [PUBMED.]
- 15 Min, K., Lee, J,. So., K., and Kim, M. "Association Between Passive Smoking and the Risk of Cervical Intraepithelial Neoplasia 1 in Korean Women." 2018. J Epidemiol. 28(1): 48-53. [PUBMED.]
- 16 Palefsky J. Biology of HPV in HIV infection.Adv Dent Res. 2006 Apr 1;19(1):99-105. Review. [PUBMED]
- 17 Abraham, A., Strickler, H., Jing, Y., Gange, S., Sterling, T., Silverberg, M., Saag, M., Rourke, S., Rachlis, A., Napravnik, S., Moore, R., Klein, M., Kitahata, M., Kirk, G., Hogg, R., Hessol, N., Goedert, J., Gill, M., Gebo, K., Eron, J., Engels, E., Dubrow, R., Crane, H., Brooks, J., Bosch, R., D’Souza, G., and North American AIDS Cohort Collaboration on Research and Design (NA-ACCORD) of IeDEA. "Invasive cervical cancer risk among HIV-infected women: A North American multi-cohort collaboration prospective study." 2013. J Acquir Immune Defic Syndr. 62(4): 405-413. [PUBMED.]
- 18 Titus-Ernstoff L, Troisi R, Hatch EE, Wise LA, Palmer J, Hyer M, Kaufman R, Adam E, Strohsnitter W, Noller K, Herbst AL, Gibson-Chambers J, Hartge P, Hoover RN. Menstrual and reproductive characteristics of women whose mothers were exposed in utero to diethylstilbestrol (DES). Int J Epidemiol. 2006 Aug;35(4):862-8. Epub 2006 May 24. [PUBMED]
- 19 Cortessis VK, Barrett M, Brown Wade N, Enebish T, Perrigo JL, Tobin J, Zhong C, Zink J, Isiaka V, Muderspach LI, Natavio M, McKean-Cowdin R. Intrauterine Device Use and Cervical Cancer Risk: A Systematic Review and Meta-analysis. Obstet Gynecol. 2017 Nov 3. doi: 10.1097/AOG.0000000000002307. [PUBMED]
- 20abc Petignat P, Roy M. "Diagnosis and management of cervical cancer." BMJ. 2007 Oct 13;335(7623):765-8 [PUBMED]
- 21 American Cancer Society Recommendations for Cervical Cancer Screening. Released March 2012. Site accessed 1-3-2020 [ACS Cervical Cancer Screening Guidelines]
- 22 Ronco G, Dillner J, Elfström KM, Tunesi S, Snijders PJ, Arbyn M, Kitchener H, Segnan N, Gilham C, Giorgi-Rossi P, Berkhof J, Peto J, Meijer CJ; the International HPV screening working group. Efficacy of HPV-based screening for prevention of invasive cervical cancer: follow-up of four European randomised controlled trials. Lancet. 2013 Nov 1. pii: S0140-6736(13)62218-7. [Epub ahead of print]
- 23ab NCCN Practical Guidelines in Oncology v.1.2008 10/26/2007 Copyright National Comprehensive Cancer Network Website accessed 12/04/07 [http://www.nccn.org/professionals/physician_gls/PDF/cervical.pdf]
- 24ab American Cancer Society Detailed Guide: Cervical Cancer. Accessed 08/03/2010 [http://www.cancer.org/docroot/CRI/CRI_2_3x.asp?dt=8]
- 25 NCCN Practice Guidelines in Oncology Cervical Cancer Screening v.1.2008 9/17/07 Accessed 12/04/07 [http://www.nccn.org/professionals/physician_gls/PDF/cervical_screening.pdf]
- 26 American Cancer Society Recommendations for Cervical Cancer Screening. Released March 2012 [http://www.cancer.org/Cancer/news/new-screening-guidelines-for-cervical-cancer]
- 27 Naucler P, Ryd W, Törnberg S, Strand A, Wadell G, Elfgren K, Rådberg T, Strander B, Forslund O, Hansson BG, Rylander E, Dillner J. Human papillomavirus and Papanicolaou tests to screen for cervical cancer. N Engl J Med. 2007 Oct 18;357(16):1589-97. [PUBMED]
- 28 Wright TC Jr, Massad LS, Dunton CJ, Spitzer M, Wilkinson EJ, Solomon D; for the 2006 American Society for Colposcopy and Cervical Pathology-sponsored Consensus Conference. 2006 consensus guidelines for the management of women with cervical intraepithelial neoplasia or adenocarcinoma in situ. J Low Genit Tract Dis. 2007 Oct;11(4):223-39. [PUBMED]
- 29 Benedet, JL. Staging Classifications and Clinical Practice Guidelines for Gynaecological Cancers. International Journal of Gynecology and Obstetrics, 70 (2000) 207-312. (pg 37-62) [http://www.figo.org/docs/staging_booklet.pdf]
- 30 PJ Eifel "Concurrent chemotherapy and radiation therapy as the standard of care for cervical cancer." Nat Clin Pract Oncol. 2006 May;3(5):248-55. [PUBMED]