
La epigenética es un campo dentro del estudio del cáncer que está creciendo con velocidad. Esta página trata algunas de las partes básicas de la epigenética y el enlace entre los cambios epigenéticos y el cáncer.
- Introducción a la epigenética
- Modificaciones epigenéticas
- Cambios epigenéticos fuera de los genes
- Causas para las mutaciones epigenéticas
- Cambios en la metilación del ADN vistos en el cáncer
- Cambios en la modificación de las histonas vistos en el cáncer
- La epigenética y la metástasis del cáncer
- La epigenética y la prevención del cáncer
- La epigenética y la detección del cáncer
- Las proteínas epigenéticas como bio-marcadores del cáncer
- Tratamientos del cáncer concentrados en la epigenética
- Medicamentos del cáncer concentrados en las histona-deacetilasas(HDACS)
- Medicamentos del cáncer concentrados en las ADN-metiltransgerasas (DNMT)
- Combinaciones de la epigenética con medicamentos del cáncer
- Limitaciones de medicamentos para el cáncer concentrados en la epigenética y direcciones futuras
Introducción a la epigenética
La base biológica del cáncer aun se desconocía hasta que se encontró una conexión entre la enfermedad y genes defectuosos. Se reveló que un cambio dentro de una secuencia específica de ADN puede provocar la reproducción (división celular) descontrolada de una célula, como se observa en el cáncer.1 Posteriormente a estos hallazgos, se descubrieron dos grupos importantes de genes asociados con el cáncer. El primer grupo constituye los oncogenes, que causan el cáncer cuando se están más activos, y el segundo grupo incluye a los genes supresores tumorales. Los supresores tumorales normalmente bloquean al cáncer, pero pueden impulsar a la enfermedad cuando son alterados o eliminados. Recientemente, se ha encubierto que el cáncer también puede ser el resultado de mutaciones epigenéticas, pequeños cambios químicos que alteran la actividad de los genes sin cambiar las secuencias de ADN en sí.
Casi todas nuestras células corporales contienen las mismas secuencias en el ADN, sin embargo está extremadamente claro que todas nuestras células no se parecen ni se comportan igualmente. Las células del corazón se ven y funcionan de diferente manera en comparación a las que están en los pulmones, aunque tengan el mismo ADN. La razón por la cuál la actividad celular varia entre células se explica con la epigenética. Epi es un prefijo griego, e indica algo que esté "arriba". Los cambios epigenéticos sirven como mecanismos para el control y la regulación de la actividad celular sin alterar a los genes permanentemente. El control epigenético depende de cambios pequeños y reversibles al ADN y a proteínas que constituyen los cromosomas. Para lograr entender la epigenética, es necesario entender la naturaleza del ADN. El ADN está compuesto por cuatro tipos de componentes químicos (nucleótidos). Los nucleótidos comparten algunas partes entre sí, pero cada uno tiene un componente único, denominado la 'base' - las bases en el ADN son la guanina (G), la citosina (C), la adenina (A) y la timina (T). El ADN tiene una forma de una escalera en espiral, con pares de bases en la mitad. Dentro de las células, el ADN no flota libremente, pues está organizado en cromosomas - estructuras donde el ADN se envuelve alrededor de unas proteínas llamadas histonas. 2 El ADN está empaquetado de tal manera por varias razones. Primeramente, las moléculas del ADN son muy, muy largas - ¡si no se las compacta, el ADN de una sola célula mediría aproximadamente 2 metros!3 Sería casi imposible que el ADN quepa dentro de una célula sin estar apropiadamente organizado. Además, la actividad de los genes se puede controlar estrictamente para que los genes estén activos solo cuando sea necesario.
¿Pero cómo es que las células logran activar a un gen que necesita? Primeramente, la porción del ADN que contiene el gen necesita desenvolverse, a través de modificaciones a las histonas. Esto se realiza con enzimas que agregan o remueven pequeños marcadores químicos, provocando que las histonas se suelten del ADN. Cuando el ADN está disponible, varias proteínas se unen al gen de interés y utilizan la información codificada. Este proceso se controla con precisión ya que la actividad no regulada de los genes en las células puede causar varios problemas, incluyendo el desarrollo del cáncer.
Modificaciones epigenéticas
As stated above, epigenetic control depends on small chemical changes to DNA or the proteins in chromosomes. There are several types of epigenetic modification. The major ones are described below.
1. Modificaciones en el ADN
La modificación más común en el ADN es la metilación. La metilación es la agregación de un pequeño grupo químico (denominado grupo metilo (-CH3)) a ciertas bases. Estas bases se modifican con metiltransferasas (DNMTs), un grupo de enzimas. La base que se altera con más frecuencia es la citosina. La incorporación de muchos grupos metilos normalmente resulta en la desactivación de un gen (silenciamiento). Existen varias maneras con las que la agregación de estos pequeños grupos logran apagar a un gen. Primero, los grupos metilos en el ADN pueden reclutar a proteínas que se unen al ADN y bloquean la activación del gen.1 Los genes no se pueden 'leer' si las proteínas adecuadas no les se pueden unir. La metilación del ADN, como las modificaciones en las histonas, se controlan con precisión, pues los problemas surgen cuando padrones normales de la metilación del ADN se alteran. Se ha descubierto que algunas células tumorales pueden estar sub-metiladas, y la cantidad de metilación disminuye en varios genes como resultado.4 Ya que la metilación se asocia con la actividad de los genes, cambios en la metilación de genes singulares también pueden provocar el cáncer. Los genes que impulsan la reproducción celular pueden activarse en exceso, o los genes que normalmente previenen el crecimiento descontrolado de las células se pueden apagar.
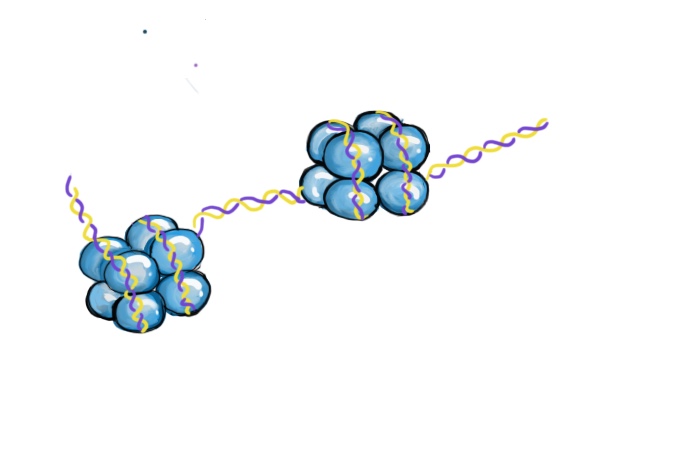
ADN envuelto alrededor de grupos de histonas. Cada grupo de 8 histonas es un nucleosoma.
2. Modificación en las histonas
DNA is organized in structures called nucleosomes, which resemble beads on a string. Each nucleosome is composed of DNA wrapped around proteins called histones. There are 8 histone proteins in each nucleosome (2 copies each of histones H2A, H2B, H3, and H4).2 The histones are arranged in the nucleosomes so that the ends of the proteins (their “tails”) stick out from the center of the structure. The protein tails are a main site of histone modification. Like all proteins, histones are made up of amino acids. Amino acids located in the tail of the histones are targets for enzymes that attach or remove chemical markers. In particular, the amino acids lysine and serine are common targets of modification.
Compared to DNA methylation, histone modification is a relatively complicated process. DNA methylation involves only two types of enzymes, those that add methyl groups to cytosine and those that remove them. There are three different types of histone modification and each has multiple types of associated enzymes. The three types of histone modifications:
- Histone phosphorylation: The addition of phosphate groups to amino acids in the histone proteins.
- Histone methylation: The most complex of the three types. Methyl groups are added to amino acids in the histone proteins. More than one kind of amino acid can be modified, and individual amino acids can have more than one methyl group added. The complexity of these steps is reflected in the many enzymes associated with histone methylation. Each addition or removal process requires a specific enzyme.
- Histone acetylation: The addition or removal of an acetyl group to the amino acid lysine. Often found on the tails of histones. Histone acetylation causes the histone to become less positively charged, and the attraction between the histone and its bound DNA is weakened.5 This results in increased gene activity because the DNA is looser and more available.
The small changes described above alter the shape of nucleosomes and change the way DNA interacts with that particular “bead”. Histone modification is a very complex process and is tightly controlled. When histone modification patterns are altered, it can lead to unregulated activity or silencing of genes. Eventually, this can lead to the death of that cell, or more seriously, to cancer.
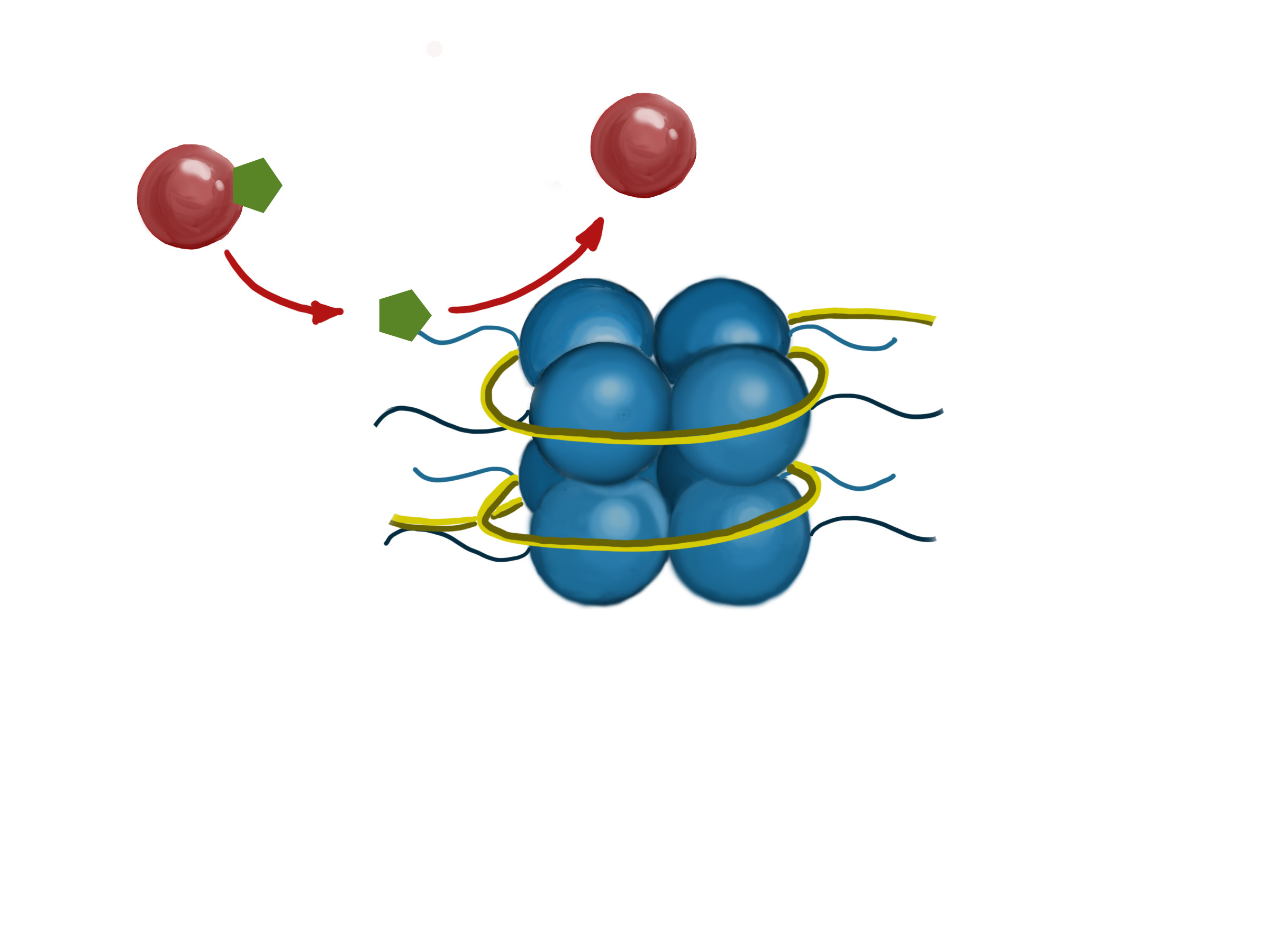
The diagram shows an enzyme (red ball) adding a chemical group (green hexagon) to the tail of a histone protein.
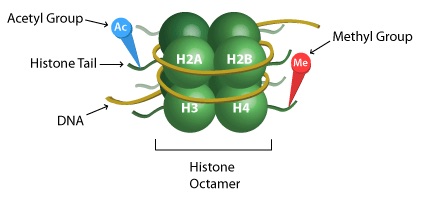
A more detailed view of nucleosome structure, showing targets of methylation and acetylation on the histone 'tails'
Cambios epigenéticos fuera de los genes
It is now understood that epigenetics plays a role in the development of cancer (carcinogenesis). As detailed above, abnormal epigenetic modifications in specific oncogenes and tumor suppressors genes can result in uncontrolled cell growth and division. However, abnormal epigenetic modifications in regions of DNA outside of genes can also lead to cancer. Protein-coding genes make up a small portion of our DNA. Humans have approximately 20,000 genes. The DNA occupied by our genes takes up less than 10% of all our DNA.6 The remaining 90% is made up of non-coding sequences. The non-coding sequences include regulatory sequences and regions of DNA that serve to provide structure to chromomes. Changes in these regions have been linked to several diseases, including cancer. It is important to note that epimutations that occur in a cell are passed on to the daughter cells formed when that cell reproduces by mitosis.
Causas para las mutaciones epigenéticas
What causes abnormal epigenetic modifications? The environment and human behavior are main causes. Poor diet, lack of exercise, drugs, exposure to environmental chemicals or radiation – these all have the potential to cause epimutations which can lead to cancer. Smoking cigarettes, for example, has been shown to affect DNA methylation patterns across multiple organ systems. The affected genes have been linked to several major diseases including cancer, osteoporosis, chronic obstructive pulmonary disease (COPD), cardiovascular disease, and rheumatoid arthritis.7 In addition behavior and environment, the set of genes a person has definitely plays a role.
A very important fact about epigenetics is that the changes to genes can be passed down from parent to child. The term for this is “transgenerational inheritance”. In other words, some epigenetic effects are inherited. One of the first studies to show this transgenerational epigenetic inheritance was done in mice. In the study, a exposing a parent to a specific odor affected the behavior and sensory neurons in later generations.8 This study was groundbreaking because it showed that we can be affected by the experiences of our parents and grandparents. The exact mechanism behind this remains unclear.
Transgenerational inheritance was thought to be impossible due to something called germline reprogramming.9 This is a process in which the DNA of reprouctive cells (sperm and egg cells) is “reset” and all epigenetic marks and modifications are erased. In theory, this would eliminate the possibility of transfer of epigenetic events between generations. However, this process isn’t as complete as we once believed. Researchers identified the DNA methylation spots in germ cells and found that although most of the genome does get demethylated, a significant number of genes retain their epigenetic marks.9 Why this happens is up for debate, but some researchers think that keeping some parental epigenetic marks may increase the offspring’s chance of survival. A practical result of this new knowledge is that future parents need to consider the possible impact of their behaviors (diet, tobacco and alcohol use, etc.) on their unborn children.
Genetics is a major factor in determining someone’s risk of developing cancer. People with certain genetic mutations carry a relatively high risk of developing cancer during their lifetimes and these mutations can often be passed on to offspring. It is thought that about one-in-twenty to one-in-ten (5%-10%) of all cancers are due to inherited mutations.10 Now that we know epigenetic changes can also be inherited, those types of changes should be considered in future studies of cancer risk.
Los cambios en la metilación del ADN vistos en el cáncer
Cancer cells often have a different epigenome, or epigenetic profile, than normal cells. DNA with less than normal amounts of DNA methylation are said to be hypomethylated. DNA with more methylation is said to be hypermethylated. A cancer cell’s epigenetic profile is typically characterized by decreased methylation across much of the genome (global DNA hypomethylation).11 The decreased methylation affects the activity of large numbers of genes. Because methylation is associated with decreased gene activity, the overall effect of hypomethylation is to increase the activity of the affected genes. If genes involved in cell growth have decreased methylation, the increased activity, and resulting cell division can lead to the development of cancer. As noted, DNA methylation changes do not have to be within protein-encoding genes to be important. Changes to DNA sequences that function as gene regulators can also cause problems.
Although the DNA of cancer cells is most commonly hypomethylated, the opposite can also be true.12 DNA hypermethylation in cancer cells tends to be limited to very specific regions (‘hot spots’). This sites affected vary by cancer type.The effect of increased DNA methylation is the opposite of hypomethylation. Hypermethylated genes tend to show decreased activity. DNA hypermethylation in cancer cells is frequently found at tumor suppressor genes; genes that function to repair DNA and control cell division. When tumor suppressor genes are silenced by increased methylation, the decrease in their activity can result in cancer development.
How do these small changes affect cancer growth? Cells with hypermethylated tumor suppressor genes are likely to grow faster than cells with normally methylated tumor suppressor genes. However, this doesn’t explain why specific sequences are found to be hypermethylated in some cancer cells but not others. The exact reason and mechanisms behind this is still unclear but, it may have something to do with how cell populations evolve over time. It is likely that the hypermethylation occurs to many genes in different cells. SOME of those changes will give those cells an advantage. Those cells will reproduce more rapidly, and take over the population.13 A good example is BRCA1, a gene linked to breast cancer, which is often found hypermethylated in breast and ovarian tumors but unmethylated in other types of tumors.14
Los cambios en las modificaciones de las histonas vistas en el cáncer
Changes to the epigenetic modifications of histones also play an important role in the development of cancer. As previously mentioned,modifications of these proteins alter the interactions between the histones and the DNA. This changes the shape of the DNA-histone complexes (nucleosomes), and alters the way that other proteins can interact with the DNA.
The epigenome of cancer cells is typically marked by a loss of histone acetyl markers, due to increased histone deacetylation. The process of histone deacetylation is catalyzed by enzymes known as histone deacetylases or HDACs. As expected, increased activity of HDACs has been found in different types of cancer cells, making HDACs an important target of epigenetic cancer treatments.15
Histone methylation is also found to be affected in cancer cells. Histone methyltransferases (HMTs) are enzymes that carry out the addition of methyl groups to histones, while histone demethylases (HDMs) have the opposite function. In cancer cells, HMTs may be altered so that they are placing methyl groups at the wrong spot, often silencing tumor suppressor genes. HDMs can also be similarly affected, leading to increased activity of oncogenes. As previously mentioned, histone methylation is very complex. The effect of methylation on gene activity can differ depending on the specific amino acid affected. As such, methyl marks on histones are categorized as either activating or repressing, depending on their effect on gene activity. An additional complication is that some HDMs have been found to be able to remove both activating and repressing marks. This poses a challenge for people developing epigenetic therapies to target HDMs - their functions must be fully understood in order to know how the drugs will affect the cancer cells.
La epigenética del metástasis del cáncer
The metastasis of cancer refers to the spread of the original (primary) tumor to a distant location in the body. Metastasis is a multi-step process: the cells must separate from the primary tumor, travel through the body to a new site through blood vessels or lymph vessels, reach a distant location, and then finally colonize the distant location to form a secondary tumor.16 Proteins have been identified that work to block cancer spread. These metastasis suppressors, which can inhibit any step of the process of metastasis. Metastatic cancer cells have been shown to epigenetically silence metastasis suppressors, often by hypermethylating these genes.17
The reasons why cancer cells metastasize is still not completely understood. Comparisons of the DNA sequences from metastatic cells and primary tumor cells were not always able to identify DNA sequence changes that could explain the difference between the cells. In 2017, researchers found an epigenetic basis for metastasis in at least one experimental model. This study examined pancreatic cancer cells from diseased patients and found that there were significant changes in the epigenome of metastatic cells, particular epigenetic changes that affected genes involved in cell migration.18
La epigenética y la prevención del cáncer
The main preventable causes of epimutations linked to cancer are environment exposures and behavior. Elimination or reduction of exposure to carcinogenic chemicals such as those found in tobacco products would likely reduce epimutations and related cancers. Other chemicals and drugs have also been found to cause epimutations, notably alcohol, which causes both DNA methylation and histone modification.19 A diet high in cruciferous vegetables, such as broccoli, cabbage, cauliflower, and kale, has been linked to a lower risk of developing prostate cancer.20 This is thought to be due to the action of several plant chemicals (phytochemicals), including sulforaphane and diindolylmethane (DIM). When researchers exposed prostate cancer cells to DIM, HDAC activity was reduce and increased amounts of the p21 tumor suppressor protein were produced.
La epigenética y la detección del cáncer
Current methods for detecting cancer use imaging techniques (X-rays, PET scans, ultrasound, etc.) or direct samples of suspicious areas to detect tumor growth. These methods are useful in detecting cancer; however, many of them rely on the detection of abnormal growths which are already present. Epigenetic detection methods are being designed to surpass the sensitivity and specificity of current tests.
Methylation-specific PCR (MSP) is an epigenetic test to identify abnormal methylation patterns in DNA. Because cancer cells are characterized by abnormal DNA methylation, it should be possible to identify cancer-specific patterns. This analysis relies on bisulfite sequencing, a technique that can distinguish between cytosines which have been methylated from those which have not been altered. MSP is useful because of its sensitivity – it can detect a single tumor cell among tens of thousands of cells.21 Another benefit is that MSP, unlike biopsies, is non-invasive. MSP can detect cancer DNA methylation patterns in plasma, stool, sputum, and urine samples.
Liquid biopsies are a cancer detection that relies on the analysis of blood samples. Liquid biopsies rely on the presence of tumor cells and/or tumor DNA in the bloodstream. These can be analyzed for both DNA mutations and epigenetic changes.22 Liquid biopsies are used in a rapidly expanding number of diseases, including cancer. There are some drawbacks to the use of liquid biopsies for the detection of epigenetic changes seen in cancer. Of upmost importance is our limited knowledge of the epimutations linked to cancer. Because different types of cancers are associated with different epimutations, it is difficult to design a general test to detect cancer. Many researchers are working to identify and categorize epigenetic patterns linked to cancer, but the process is slowed by the fact that each identified change must be confirmed in many patients.
Las proteínas relacionadas a la epigenética como biomarcadores del cáncer
A biomarker is an indicator of a condition that is not easily directly measured. Examples of biomarkers include the use of blood cholesterol levels and blood pressure as indicators of heart disease. Most current research into epigenetic cancer detection methods relies on identifying and detecting specific epigenetic changes. Ideally, it would be great to identify a set of changes (or a protein) that is present in most or all types of cancer. A 2017 study has identified a protein which may serve as just such a marker. UHRF1 is a protein encoded by the UHRF1 gene. It binds to specific DNA sequences where it recruits DNMT1, a DNA methyltransferase. It also functions in the coordination of different epigenetic enzymes, such as DNMTs and HDACs, to maintain normal DNA methylation patterns and histone modification patterns.23 UHR1 has been shown to be highly active in many types of cancer which makes it a potential universal cancer biomarker.24 UHRF1 has been identified as a protein that promotes the development of cancer. Increased levels of UHRF1 causes global DNA hypomethylation by destabilizing DNMTs. Increased UHRF1 activity have been linked to tumor growth, metastasis, and resistance to radiotherapy in different types of cancer, including lung, liver, breast, pancreatic, colorectal, prostate, and kidney (renal). High levels of UHRF1 have also been linked to lower survival rates, increased resistance to therapy, and increased rates of recurrence. Abnormal UHRF1 activity can be detected in the early stages of tumor development, making it a good candidate for early diagnosis of cancer. Targeting UHRF1 may also improve the effectiveness of radiotherapy.
Tratamientos del cáncer concentrados en cambios epigenéticos
Most current cancer treatments, including radiotherapy and chemotherapy, are cytotoxic treatments. Their goal is to kill cancer cells. However, these treatments are limited in their effectiveness as they often damage or kill normal cells, and drug resistance frequently develops in the cancer cells. Due to these limitations, other types of treatments are being researched, including immunotherapies, and epigenetic treatments.
Epigenetic treatments or “epi-drugs” refer to drugs that reverse abnormal epigenetic modifications in cancer cells. The main benefit of epi-drugs is that they are not cytotoxic, their goal is to “reprogram” cancer cells.25 Unlike genetic mutations, epimutations are reversible, which allows for the possibility to reverse the epimutations in cancer cells. The targets of epigenetic therapy are the enzymes involved in epigenetic modifications, including HDACs, DNMTs and HDMs. Although any type of epigenetic enzyme is a potential target for epigenetic therapy, research has focused on enzymes that are less complex and well-studied. The two main types of epi-drugs being investigated are HDAC inhibitors and DNMT inhibitors.
Inhibidores de la histona-deacetilasa (HDAC)
Histone acetylation is the process by which enzymes add acetyl groups to histones. The result is often an increase in gene activity. The reverse process, histone deacetylation, has the opposite effect, leading to gene silencing. In many types of cancer cells, high histone deacetylase activities have been observed. The increase is linked to the silencing of tumor suppressor genes and DNA repair genes. The aim of HDAC inhibitors is to decrease HDAC activity and indirectly increase the activity of tumor suppressors.
Many HDAC inhibitors are currently being researched and several of these have been approved for clinical use including vorinostat (Zolinza®), romidepsin (Istodax®), bellinostat (Beleodaq®), and panobinostat (Farydak®).26 These epi-drugs work by binding to HDACs, blocking their ability to remove acetyl groups from histones. These four drugs are pan-HDAC inhibitors - they target most or all HDACs. Although the approval of HDAC inhibitors is a step in the right direction, the epi-drugs have some limitations and disadvantages. One is that these drugs have only shown to be effective for treating blood (hematologic) cancers, including lymphoma, leukemia, and myeloma. Another drawback is that the drugs have side effects including fatigue and diarrhea. They are also toxic to bone marrow, and can reduce blood cell counts . The side effects are at least party due to their lack of specificity. By targeting all types of HDACs. They cause side effects by inhibiting HDACs (or other enzymes) necessary for normal cellular function. HDAC inhibitors designed to specifically target well-studied HDACs, could have better results and less side effects.
Inhibidores de la ADN-metiltransferasa (DNMT)
As discussed above, DNA methylation is the process in which enzymes called DNA methyltransferases add methyl groups to bases in DNA. Because these changes affect the activity of the genes, DNA methylation plays a role in cancer development. Tumor suppressor genes are often heavily methylated and silenced. DNMT inhibitors have therefore been studied for their possible use as epi-drugs.27 In theory, DNMT inhibitors can reverse gene silencing and restore normal tumor suppressor function.
Two DNMT inhibitors have been shown to be effective as anticancer drugs and are currently approved for use in treating acute myeloid leukemia and myelodyplastic syndrome: azacytidine (Vidaza®) and decitabine (Dacogen®). When these drugs enter cells, they interact with DNA and inhibit any DNMTs that come along. The bound DNMT is unable to further methylate other regions of DNA and is ultimately destroyed. Although both epi-drugs are widely used in clinical treatment, they have several disadvantages. One drawback is their chemical instability; they are rapidly broken down, often in less than an hour and the drugs are changed into inactive compounds. Another downside is the toxicity of the drugs. They can cause DNA damage and lower immune function.
A new experimental DNMT inhibitor, zebularine, has been shown to be effective. Although it has not yet reached clinical trial stages, it is promising because it may improve upon the drawbacks of azacytidine and decitabine. Zebularine inhibits DNMTs and reverses gene silencing in cancer cells. It does so in a similar method to azacytidine and decitabine by interacting with DNA.28 When DNMTs encounter DNA with zebularine stuck to it, a a very stable complex is formed,’trapping’ the DNMT and blocking methylation. Zebularine offers several advantages over the two approved DNMT inhibitors. It is more stable in the body and it appears to be much less toxic.
Combinaciones con terapias epigenéticas
Complex diseases such as cancer often prove difficult to treat. Cancers also frequently develop resistance to any individual treatment. Combination therapy has emerged as a method to treat cancer more effectively. Combination therapy refers to the use of multiple treatments at the same time. For cancer patients, this often involves some combination of surgery, epigenetic drugs, radiotherapy, chemotherapy, and targeted therapies.
The combination of epigenetic drugs and radio/chemotherapy appears to be useful as each therapy may be able to compensate for the other’s disadvantages. Radiation and chemotherapy are effective in killing cancer cells and slowing growth; however, these treatments are often not be enough on their own. If all the cancer cells are not killed, then recurrence is possible. Combination treatments that include epigenetic drugs may increase sensitivity to chemotherapy and radiation therapy. 29
Combination epigenetic therapy is also a possibility. This involves the use of two or more different types of epigenetic drugs, including HDAC inhibitors and DNMT inhibitors. A significant benefit to combined epigenetic therapy is that it allows the use of lower drug doses. This could reduce side effects and increase effectiveness. Research with acute myeloid leukemia cells showed that combined decitabine-vorinostat treatment resulted in increased cancer cell death.30 Combining HDAC and DNMT inhibitors can result in increased histone acetylation compared to the use of HDAC inhibitors alone. The increase can be explained by the “communication” that occurs between DNA methylation and histone modification. The effect of DNA methylation is boosted by histone modification. Proteins that bind to methyl groups on DNA have the ability to recruit other enzymes, including HDACs, further decreasing gene activity.31
Limitaciones y direcciones futuras
Current epigenetic therapy is promising; however, several obstacles must be overcome to make it more effective. Although epigenetic drugs have shown to be effective against hematological cancer, a major limitation is their ineffectiveness against solid tumors. This is largely because of the environment of tumors typically have very low oxygen levels (low oxygen=hypoxia).32 Hypoxia occurs in tumors due to the presence of abnormal blood vessels, which cannot provide rapidly growing cancer cells with enough oxygen. The conditions alter the behavior of the tumor cells. Some arise which are characterized by aggressiveness, metastasis, and resistance to chemotherapy and radiation. Hypoxia results in the production of a different group of enzymes and proteins that take over control of gene activity. Additionally, tumor cells living in hypoxic conditions have an unusual epigenetic profile characterized by a decrease in active histone marks, an increase in repressive histone marks, and decreases in histone acetylation and DNA methylation. This is significant because current HDAC and DNMT inhibitors do not seem to be effective against tumor cells growing in hypoxic conditions. Research is required to better understand the epigenetic patterns of cancer cells living in hypoxic conditions. This information will help us to develop epigenetic drugs to target solid tumors.
Combination therapy can also be improved. This treatment approach relies on different types of cancer therapies working together; however, extensive research must be done to discover the best combinations of different drugs, proper dosing and timing. The development of new epigenetic drugs is also critical in improving cancer treatment. One of the main objectives is to design drugs with increased specificity. Ongoing research includes further exploration of epigenetic biomarkers linked to cancer and work to understand the epigenetic mechanisms of drug resistance in cancer cells. A major reason to study cancer epigenetics is inform the development of safe, effective treatments and to help prevent and detect cancer.
- 1 a b Chen, Q. W., Zhu, X. Y., Li, Y. Y., & Meng, Z. Q. (2014). Epigenetic regulation and cancer. Oncology Reports, 31, 523-532. [PUBMED]
- 2 a b Kouzarides, T. (2007). Chromatin Modifications and Their Function. Cell, 128, 693-705. [PUBMED]
- 3Annunziato, A. (2008) DNA Packaging: Nucleosomes and Chromatin. Nature Education 1(1):26. [View]
- 4Kulis, M., & Esteller, M. (2010). 2 – DNA Methylation and Cancer. Advances in Genetics, 70, 27-56. [PUBMED]
- 5Bannister, A. J., & Kouzarides T. (2011). Regulation of chromatin by histone modifications. Cell, 21(3), 381-395. [PUBMED]
- 6Powledge, T. M. (2014, August 12). How much of human DNA is doing something? Retrieved July 26, 2017 [View]
- 7Joehanes, R., et al. (2016). Epigenetic Signatures of Cigarette Smoking. Circulation: Cardiovascular Genetics, 9(5), 436-447. [PUBMED]
- 8Dias, B. G., & Ressler, K. J. (2013). Parental olfactory experience influences behavior and neural structure in subsequent generations. Nature Neuroscience, 17, 89-96. [PUBMED]
- 9 a b Heard, E., & Martienssen, R. A. (2014). Transgenerational Epigenetic Inheritance: myths and mechanisms. Cell, 157(1), 95-109. [PUBMED]
- 10Genetic Testing for Hereditary Cancer Syndromes. (2013, April 11). Retrieved July 14, 2017. [View]
- 11Sharma, S., Kelly, T. K., Jones, & P. A. (2010). Epigenetics in cancer. Carcinogenesis, 31(1), 27-36. [PUBMED]
- 12De Bustros, A., Nelkin, B. B., Silverman, A., Ehrlich, G., Poiesz, B., & Baylin, S. B. (1988). The short arm of chromosome 11 is a "hot spot" for hypermethylation in human neoplasia. Proc Natl Acad Sci USA, 85(15), 5693-5697. [PUBMED]
- 13Esteller, M. (2002). CpG island hypermethylation and tumor suppressor genes: a booming present, a brighter future. Oncogene, 21(35), 5427-5440. [PUBMED]
- 14Esteller, M., et al. (2000). Promoter hypermethylation and BRCA1 inactivation in sporadic breast and ovarian tumors. J Natl Cancer Inst, 92(7), 564-569. [PUBMED]
- 15Halkidou,K., et al. (2004). Upregulation and nuclear recruitment of HDAC1 in hormone refractory prostate cancer. Prostate, 59, 177–189. [PUBMED]
- 16Mandal, A. (2014, October 08). What is Metastasis? Retrieved July 28, 2017 [View]
- 17Li, Q. & Chen, H. (2011). Epigenetic modifications of metastasis suppressor genes in colon cancer metastasis. Epigenetics, 6(7), 849-852. [PUBMED]
- 18McDonald, O. G. et al. (2017). Epigenetic reprogramming during pancreatic cancer progression links anabolic glucose metabolism to distant metastasis. Nature Genetics, 49, 367-376. [PUBMED]
- 19Mahnke, A. H., Miranda, R. C., & Homanics, G. E. (2017). Epigenetic mediators and consequences of excessive alcohol consumption. Alcohol, 60, 1-6. [PUBMED]
- 20Beaver, L. M., et al. (2012). 3,3′-Diindolylmethane, but not indole-3-carbinol, inhibits histone deacetylase activity in prostate cancer cells. Toxicology and Applied Pharmacology, 263(3), 345-351. [PUBMED]
- 21Zhu, J., & Yao, X. (2008). Use of DNA methylation for cancer detection: Promises and challenges. The International Journal of Biochemistry and Cell Biology, 41, 147-154. [PUBMED]
- 22Alix-Panabières, C. & Pantel, K. (2013). Circulating Tumor Cells: Liquid Biopsy of Cancer. Clinical Chemistry, 59(1), 110-118. [PUBMED]
- 23Unoki, M., Brunet, J., & Mousli, M. (2009). Drug discovery targeting epigenetic codes: the great potential of UHRF1, which links DNA methylation and histone modifications, as a drug target in cancers and toxoplasmosis. Biochem Pharmacol, 78, 1279-1288. [PUBMED]
- 24Ashraf, W. et al. (2017). The epigenetic integrator UHRF1: on the road to become a universal biomarker for cancer. Oncotarget. [PUBMED]
- 25Ronnekleiv-Kelly, S. M., Sharma, A., & Ahuja, N. (2017) Epigenetic therapy and chemosensitization in solid malignancy. Cancer Treatment Reviews, 55, 200-208. [PUBMED]
- 26Ceccacci, E., & Minucci, S. (2016). Inhibition of histone deacetylases in cancer therapy: lessons from leukaemia. British Journal of Cancer, 114(6), 605-611. [PUBMED]
- 27Gnyszka, A., Jastrzebski, Z., & Flis, S. (2013). DNA Methyltransferase Inhibitors and Their Emerging Role in Epigenetic Therapy of Cancer. Anticancer Research, 33(8), 2989-2996. [PUBMED]
- 28Champion, C. et al. (2010). Mechanistic Insights on the Inhibition of C5 DNA Methyltransferases by Zebularine. PLoS One, 5(8), e12388. [PUBMED]
- 29Pajonk, F., Vlashi, E., & McBride, W. H. (2010). Radiation Resistance of Cancer Stem Cells: The 4 R’s of Radiobiology Revisited. Stem Cells, 28(4), 639–648. [PUBMED]
- 30Young, C. S., Clarke, K. M., Kettyle, L. M., Thompson, A., & Mills, K. I. (2017). Decitabine-Vorinostat combination treatment in acute myeloid leukemia activates pathways with potential for novel triple therapy. Oncotarget. [PUBMED]
- 31Jones, P. L., et al. (1998). Methylated DNA and MeCP2 recruit histone deacetylase to repress transcription. Nature Genetics, 19, 187-191. [PUBMED]
- 32Ramachandran, S., Ient, J., Göttgens, E., Krieg, A. J., & Hammond, E. M. (2015) Epigenetic Therapy for Solid Tumors: Highlighting the Impact of Tumor Hypoxia. Genes, 6, 935-956. [PUBMED]
