Thanks to your immune system, you already have the power to fight cancer inside of you! Although the immune system is usually associated with protecting us from viruses, bacteria, and other disease-causing organisms, it actually has a big role in helping fight cancer by being able to recognize and kill cancer cells.
A major function of the immune system is to recognize foreign objects (viruses, bacteria, parasites, splinters, and anything else that isn’t supposed to be in your body) and destroy them. Immune cells do this by recognizing specific targets on the surface of the object. Cancer cells are abnormal and may be recognized as ‘foreign’ by immune cells. Immune cells can then kill these cancer cells.
Learn more in our section on the immune system.
Topics discussed on this page:
- Why do people get cancer?
- Immune system-cancer cell battles.
- How can cancer cell defenses be overcome?
Unfortunately, the immune system is also deeply involved in the development of cancer. While this is certainly not something that is good for the person, the cells are not 'thinking' about what they are doing, and are only really doing what they are being told to do. You can learn more about that here:
Why do people get cancer?
A major function of the immune system is to recognize foreign objects (viruses, bacteria, parasites, splinters, and anything else that isn’t supposed to be in your body) and destroy them. Immune cells do this by recognizing specific targets on the surface of the item to be destroyed. Cancer cells are abnormal and may be recognized as ‘foreign’ by immune cells. Immune cells recognize small molecular ‘flags’ associated with tumor cells (called tumor specific antigens (TSAs) or tumor associated antigens (TAAs)). Immune cells can then kill these cancer cells.
Note: An antigen can be defined as anything the immune system can recognize. It is a general term. Antigens can be proteins, sugars, lipids, nucleic acids, or hybrid molecules. Most antigens are proteins.
If the immune system is able to recognize and kill cancer cells, why do people get cancer?
It is thought that most cancers are caught very early and eliminated by the immune system. The cancers that survive have to be able to evade the immune system. It turns out that there are several ways that cancer cells avoid being recognized and killed. These include:
- Cancer cells form from normal cells. Because cancer arises from a person’s own cells, it’s trickier for the immune system to recognize cancer cells than to identify a truly foreign invader like a virus.
- Cancer cells with the most targets on them are most easily killed by the immune system. This sounds like a good thing, and it can make tumors shrink, but after a while, the cancer cells that are left have fewer targets on their surface. Those with the least amount of target antigens on their surface have the best chance of surviving, and tend to take over, making the cancer resistant to immune cells. It is similar to treating a lawn with weed killer. Any weeds that are resistant will survive and grow. After that, the weed killer will not work anymore. The immune system accidentally ‘selects’ for cancer cells that it can’t recognize.
- Cancer cells can hijack normal control systems to turn the immune system ‘off’ in and around the tumor. To keep the immune system under control, there are several controls (like ‘on-off’ switches) that work to regulate the activity of immune cells. Some cancer cells are able to flip that switch, turning the immune system off in the area around and in the tumor. Treatments designed to reverse this are very promising and are described more below.
- Cancer cells can evade the immune system by making less of the ‘self’ signals that immune cells use to recognize defective or infected cells. Cancer proteins can be shown to the immune system by being stuck to cellular versions of flagpoles. By taking down the poles, cancer cells prevent this from happening and are able to avoid being recognized and killed.1 2 3
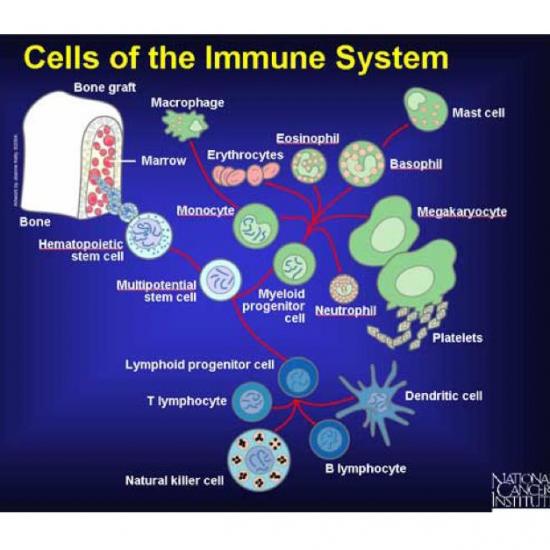
Immune system- cancer cell battles
The battle between the immune system and cancer is complex and can last for many years. It is likely that most cancers are eliminated by the immune system at very early stages, before there would be any evidence of their existence. It is only the cancers that are able to avoid the immune system that can grow to become dangerous.
How does the immune system kill cancer cells?
The immune system is divided up like a military organization, with different cells armed with different weapons to attack different kinds of targets.
Natural killer (NK) cells are able to recognize proteins on a tumor cell’s surface, They release proteins that punch holes in the target cell membrane, and cause cell death.
Other cells, (macrophages and dendritic cells) may join in, and release cell-signaling proteins (cytokines) that recruit and ‘activate’ T-cells. Stimulated T-cells can directly kill cancer cells or recruit yet another type of immune cell, B-cells. B-cells produce proteins (antibodies) that can stick to targets on the cancer cells. This further intensifies the response and can lead to killing of the cancer cells.4 5 6 7
How can a cancer cell shut down the immune system?
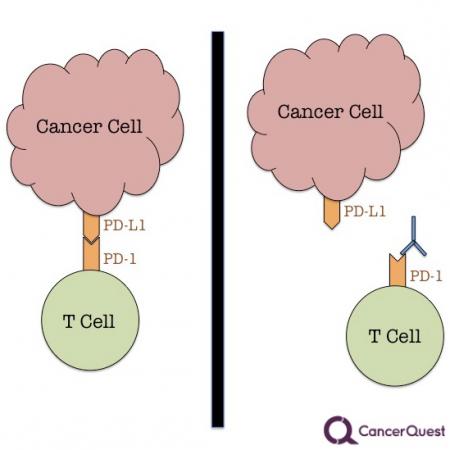
As mentioned previously, the immune system is a powerful weapon. Not surprisingly, there are controls in place to turn off the system when it is no longer needed. One ‘off switch’ is called programmed death ligand 1 (PD-L1). It is a protein that is normally found on immune cells called macrophages. PD-L1 binds to another protein, PD-1, which is found on the surface of T-cells When these two proteins come into contact with each other the T-cells are ‘told’ to stop attacking.
If a cancer cell has PD-L1 on its surface, T-cells that come into contact with that cell are likely to shut down their defensive activities. This leads to cancer cell survival.
An additional signal that tells T-cells to stop attacking cancer cells is sent by another pair of proteins, CTLA-4 and B7-1. Like PD-1, CTLA-4 is found on the surface of T cells. Like the PD-1/PD-L1 interaction, when CTLA-4 binds to its partner, the T-cell becomes inactive.8 9 10
How can cancer cell defense be overcome?
Many researchers are working to develop treatments that will ‘wake up’ and activate the immune system in cancer patients. Drugs that target immune-evasion tactics used by cancer have produced some very exciting results and are being very actively studied.
Treatments to stimulate the immune system fall into a few different categories.
1. General immune activation
There are several proteins that work to cause general immune activation. These cell to cell signals (cytokines) turn up the immune activity in the body. They are not specific to any particular target or cancer. The idea behind using these to treat cancer is that the immune system can kill cancer if it is active. These signals are like putting a military on ‘high alert’. Examples of immune stimulating cytokines are alpha-interferon (α-IFN) and interleukin 2 (IL-2). These proteins can be man-made, and are approved treatments for some forms of cancer.11
12
13
14
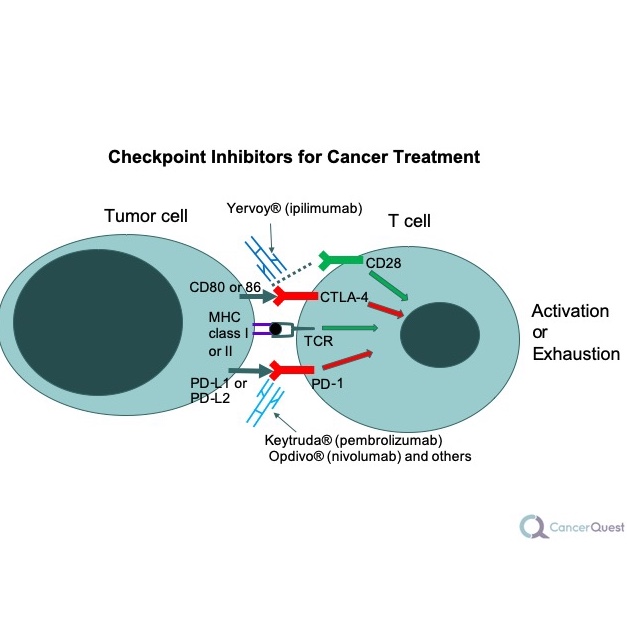
2. Checkpoint blockers
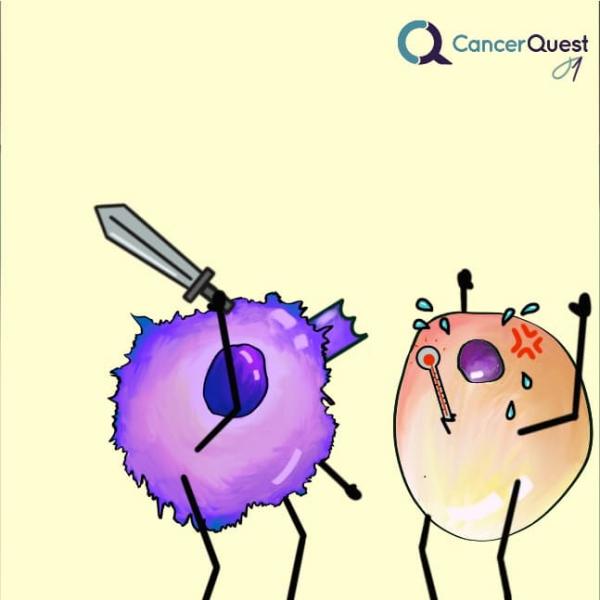
Immune cells can recognize cells that have become infected or damaged - including cancer cells.

The infected or cancerous cells can be killed by immune cells.
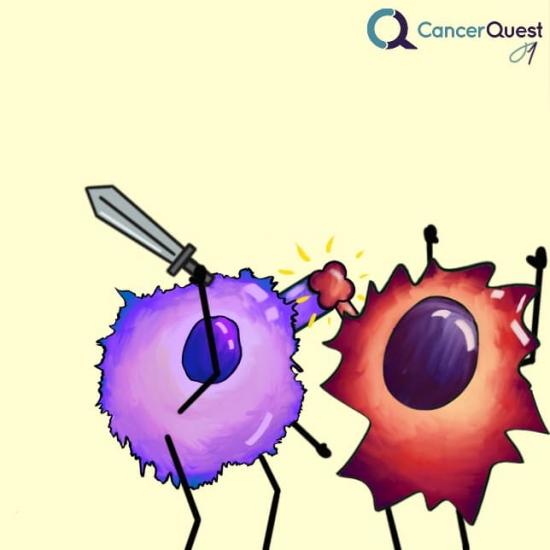
Some cancer cells can interact with immune cells in a way that inactivates the immune cells.
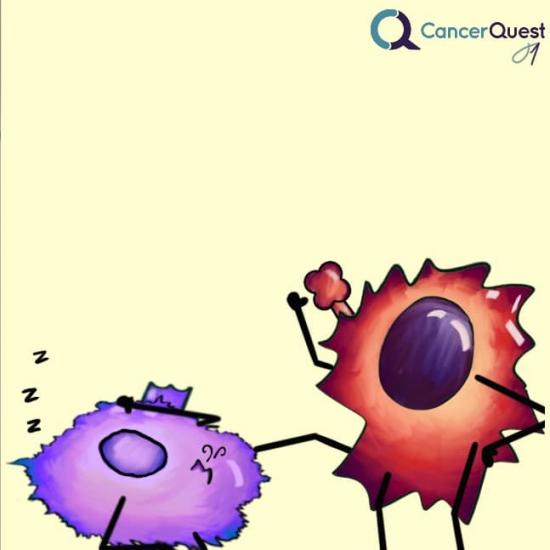
The immune cells are made inactive - like they are asleep.

These drugs work to ‘turn on’ immune cells that have been switched off by one of the protein interactions described previously.
One of the first cancer immunotherapies to become FDA-Approved is ipilimumab (brand name Yervoy®). The drug is actually a protein (an antibody). Antibodies are proteins that bind very specifically to targets, including other proteins. Antibodies neutralize the target and sometimes signal other cells to destroy the target. Yervoy helps prolong the immune response against cancer by binding to and inhibiting another protein (CTLA-4). If CTLA-4 binding to B7-1 is blocked, a more sustained immune response can occur.
Yervoy® was intially approved to treat melanoma because clinical studies of Yervoy® showed that it extended survival for melanoma patients.15 It has since been approved for some cases of kidney and colorectal cancer.16 Yervoy® is currently undergoing clinical studies to test its effectiveness in treating lung cancer , bladder cancer, prostate cancer and others17 .
Pembrolizumab (Keytruda®) is an antibody drug that binds to PD-1, blocking the interaction between PD-1 and PD-L1. This treatment is approved for use in melanoma patients 18 and received 'breakthrough therapy' designation from the FDA as a treatment for some forms of lung cancer.19 Keytruda has since been approved for several other cancer types.20
Check for clinical trials of pembrolizumab (Keytruda®) (Opens in a new window)
Another anti-PD-1 antibody, nivolumab (brand name - Opdivo®),was first approved in the U.S. for use in melanoma and non-small cell lung cancer.21 Opdivo® works like Keytruda®; by blocking the PD-1/PD-L1 interaction, it allows T cells to stay active, even if tumor cells have PD-L1 on their surface. It has since been approved to treat several other cancers and testing against more cancers is underway.22
Check for clinical trials of nivolumab (Opdivo®). (Opens in a new window.)
The anti-PD-L1 antibody atezolizumab (brand name TECENTRIQ®) works in a way similar to Opdivo®, but it sticks to PD-1’s partner, PD-L1. The results are the same, activation of the immune system. TECENTRIQ® is used to treat some kinds of bladder, lung and breast cancers.23
Check for clinical trials of atezolizumab (TECENTRIQ®). (Opens in a new window.)
The image to the right shows the interaction between a cancer cell and a T cell, a type of immune system cell. When the the PD-1/PD-L1 interaction occurs, it shuts down the activity of that T cell. This limits the ability of the body to fight cancer. The antibody drugs, prevent that from happening by sticking to one of the partners. The diagrams above and below shows an antibody stuck to the PD-1 protein. This is the way that Keytruda® and Opdivo® work.
3. Cancer vaccines
Another approach is to try to boost the immune response to cancer by using cancer cell proteins to stimulate the immune system. Individual proteins, or mixtures of proteins can be used to teach the immune cells what they should be looking for. This is very similar to the way that vaccines work to prevent diseases like mumps and measles.
There are several approaches to cancer vaccines being studied. These are covered in a separate treatment section.
Learn more about cancer vaccines.
4. Chimeric antigen receptor (CAR) T cell therapy takes the idea behind cancer vaccines one step further. Instead of activating antigen presenting cells against the person’s cancer, the person’s T cells are modified to specifically attack cancer cells. This is done by genetically modifying the patient’s T cells to recognize proteins on the surface of cancer cells.24 25 The results have been very exciting. A clinical trial of CAR T cells used against B cell acute lymphocytic leukemia (ALL) yielded an 88% overall complete response rate.26
On July 12, 2017, the FDA advisory committee unanimously voted to recommend the approval of a CAR T-cell therapy for children and young adults with acute lymphoblastic leukemia (ALL) that has relapsed or resisted treatment.27 On August 30, 2017 the FDA approved the cancer treatment Kymriah®, the first gene therapy legally available in the United States. It was approved to treat patients under the age of 25 with certain types of B-cell acute lymphoblastic leukemia (ALL).28 In October 2017, the FDA approved a second CAR T-cell therapy. Yescarta® (axicabtagene ciloleucel) was approved for some patients with diffuse large B-cell lymphoma (DLBCL).29
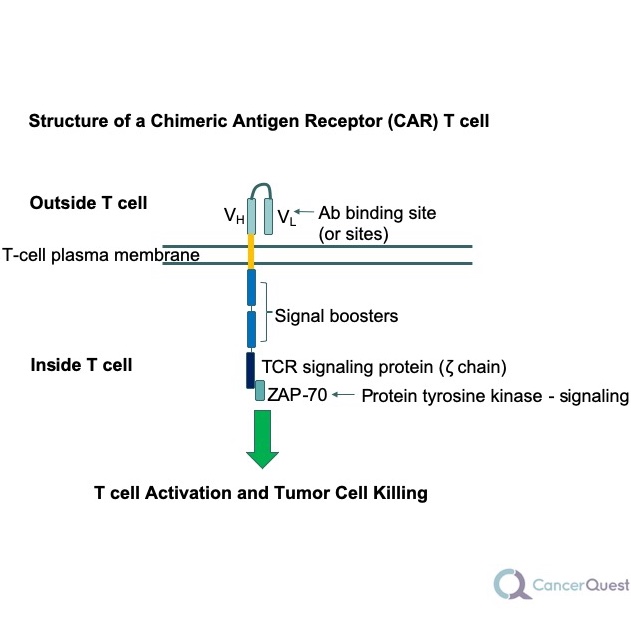
The image above shows a generalized structure of a CAR T-cell receptor. The antibody (Ab) binding site(s) are used to recognize the tumor cell and then signals are created inside the cell that cause the T cell to kill the targeted cancer cells.
- 1Martinez-Bosch, N., Vinaixa, J., and Navarro, P. "Immune evasion in pancreatic cancer: from mechanisms to therapy." Cancers (Base!) 2018; 10(1): 6. [PUBMED]
- 2Seliger B. Strategies of tumor immune evasion. BioDrugs. 2005;19(6):347-54. [PUBMED]
- 3Poggi A, Musso A, Dapino I, Zocchi MR. Mechanisms of tumor escape from immune system: role of mesenchymal stromal cells. Immunol Lett. 2014 May-Jun;159(1-2):55-72. Epub 2014 Mar 20. [PUBMED]
- 4Vesely, M. and Schreiber, R. "Cancer immunoediting: antigens, mechanisms, and implications to cancer immunotherapy." Ann N Y Acad Sci. 2013 May;1284(1):1-5. [PUBMED]
- 5Wellenstein, MD and de Visser, KE. Cancer-cell-intrinsic mechanisms shaping the tumor immune landscape. Immunity. 2018 Mar;48(3):399-416. [PUBMED]
- 6Smyth MJ, Dunn GP, Schreiber RD. Cancer immunosurveillance and immunoediting: the roles of immunity in suppressing tumor development and shaping tumor immunogenicity. Adv Immunol. 2006;90:1-50. [PUBMED]
- 7Daher, M. and Rezvani, K. "Next generation natural killer cells for immunotherapy: the promise of genetic engineering." Curr Opin Immunol. 2018;51:146-153. [PUBMED]
- 8Gong, J., Chehrazi-Raffle, A., Reddi, S., and Salgia, R. Development of PD-1 and PD-L1 inhibitors as a form of cancer immunotherapy: a comprehensive view of registration trials and future considerations. J Immunother Cancer. 2018 Jan 23; 6(1):8. [PUBMED]
- 9Zamarin D, Postow MA. Immune checkpoint modulation: rational design of combination strategies. Pharmacol Ther. 2015 Jun;150:23-32. Epub 2015 Jan 10. [PUBMED]
- 10Ribas, A. and Wolchock JD. "Cancer immunotherapy using checkpoint blockade." Science. 2018 Mar 23;359(6382): 1350-1355. [PUBMED]
- 11Rizza P, Moretti F, Belardelli F. Recent advances on the immunomodulatory effects of IFN-alpha: implications for cancer immunotherapy and autoimmunity. Autoimmunity. 2010 Apr;43(3):204-9. [PUBMED]
- 12Ferrantini M, Capone I, Belardelli F. Interferon-alpha and cancer: mechanisms of action and new perspectives of clinical use. Biochimie. 2007 Jun-Jul;89(6-7):884-93. Epub 2007 Apr 21. [PUBMED]
- 13Rosenberg SA. IL-2: the first effective immunotherapy for human cancer. J Immunol. 2014 Jun 15;192(12):5451-8 [PUBMED]
- 14Sim GC, Radvanyi L. The IL-2 cytokine family in cancer immunotherapy. Cytokine Growth Factor Rev. 2014 Aug;25(4):377-90. Epub 2014 Aug 1. [PUBMED]
- 15Hodi FS, O'Day SJ, McDermott DF, Weber RW, Sosman JA, Haanen JB, Gonzalez R, Robert C, Schadendorf D, Hassel JC, Akerley W, van den Eertwegh AJ, Lutzky J, Lorigan P, Vaubel JM, Linette GP, Hogg D, Ottensmeier CH, Lebbé C, Peschel C, Quirt I, Clark JI, Wolchok JD, Weber JS, Tian J, Yellin MJ, Nichol GM, Hoos A, Urba WJ. Improved survival with ipilimumab in patients with metastatic melanoma. N Engl J Med. 2010 Aug 19;363(8):711-23. Epub 2010 Jun 5. [PUBMED]
- 16Hematology/Oncology (Cancer) Approvals & Safety Notifications. Accessed 1-7-2020 [U.S. FDA Cancer Drug Approvals]
- 17NIH Clinical Trials Search. Accessed 1-7-2020. [Clinical Trials Search]
- 18FDA approves Keytruda for advanced melanoma. U.S. Food and Drug Administration website. Published September 4, 2014. Updated September 10, 2014. Accessed September 13, 2014. [http://www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm412802.htm.]
- 19Merck Receives FDA Breakthrough Therapy Designation for KEYTRUDA® (pembrolizumab) in Advanced Non-Small Cell Lung Cancer 11/27/2014 [http://www.mercknewsroom.com/news-release/oncology-newsroom/merck-receives-fda-breakthrough-therapy-designation-keytruda-pembroli]
- 20Drugs.com Keytruda Information Page. Accessed 1-7-2020 [Keytruda Information]
- 21Drugs.com Opdivo FDA Approval History. Accessed 1-7-2020 [Drugs.com Opdivo Approval History]
- 22Clinical Trials Search Results for Opdivo® [Search NIH Clinical Trials Database]
- 23Manufacturer's website. Accessed 1-3-2020. [TECENTRIQ website]
- 24Chakravarti D, Wong WW. Synthetic biology in cell-based cancer immunotherapy. Trends Biotechnol. 2015 Jun 15. pii: S0167-7799(15)00110-9. [Epub ahead of print] [PUBMED]
- 25Sharpe M, Mount N. Genetically modified T cells in cancer therapy: opportunities and challenges. Dis Model Mech. 2015 Apr;8(4):337-350. Epub 2015 Apr 1. [PUBMED]
- 26Davila ML, Riviere I, Wang X, Bartido S, Park J, Curran K, Chung SS, Stefanski J, Borquez-Ojeda O, Olszewska M, Qu J, Wasielewska T, He Q, Fink M, Shinglot H, Youssif M, Satter M, Wang Y, Hosey J, Quintanilla H, Halton E, Bernal Y, Bouhassira DC, Arcila ME, Gonen M, Roboz GJ, Maslak P, Douer D, Frattini MG, Giralt S, Sadelain M, Brentjens R. Efficacy and toxicity management of 19-28z CAR T cell therapy in B cell acute lymphoblastic leukemia. Sci Transl Med. 2014 Feb 19;6(224):224ra25 [PUBMED]
- 27Mika, Aggie. "FDA Votes Yes on CAR T-Cell Therapy." The Scientist (2017). [THE SCIENTIST]
- 28FDA approves Kymriah for the treatment of refractory B-cell ALL in patients under 25 years of age. [https://www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm574058.htm]
- 29FDA approves CAR-T cell therapy to treat adults with certain types of large B-cell lymphoma. FDA website. Accessed 12-1-19 [FDA website]
