Zoledronic Acid
Zometa® (zoledronic acid) is used to treat hypercalcemia of malignancy (HCM). HCM is a condition in which calcium is released into the blood stream from the bones in excess. HCM can be caused by factors (proteins) that tumors release into the blood that in turn circulate throughout the body and activate cells that degrade bone. HCM can also result from metastases (spreading of cancer cells) in bone. Solid and myelogenous cancers can cause bone loss and calcium release by stimulating bone degrading cells (osteoclasts). Zometa® is administered as an IV infusion and its administration is accompanied by other cancer fighting drugs.1
- 1 Zoledronic Acid. MedlinePlus. 2016. https://medlineplus.gov/druginfo/meds/a605023.html
Although the mechanism by which Zometa® acts is not completely understood, it is thought that there are a couple of mechanisms may be responsible for its action. Zometa® inhibits the particular cells (osteoclasts) that are responsible for degrading (breaking down) bone, and also causes apoptosis (cell death). Also, it binds to bone and directly blocks it from being broken down and released into the bloodstream.1
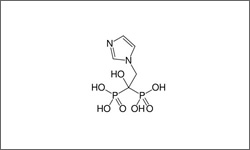
The diagram below shows the 3D molecular structure of Zoledronic Acid.
- 1 Zoledronic Acid. MedlinePlus. 2016. https://medlineplus.gov/druginfo/meds/a605023.html
Patients may occasionally experiences flu-like symptoms after infusion. It is important that patients undergo regular tests to evaluate the levels of certain important factors in the blood that may become depleted with treatment. Since Zometa® can affect the kidneys, before every infusion blood tests should be done to monitor kidney function.1
- 1 Zoledronic Acid. MedlinePlus. 2016. https://medlineplus.gov/druginfo/meds/a605023.html
