Vemurafenib
The drug is used to treat advanced or metastatic melanoma that can not be removed by surgery. It is only used in patients whose cancer cells have the BRAF V600E mutation. Vemurafenib is given as a pill, typically two pills at a time, twice daily.1
- 1 Chu, E., & DeVita, V. T. (2015). Physicians' cancer chemotherapy drug manual 2015. Burlington, MA: Jones & Bartlett Learning.
Vemurafenib (ZELBORAF®) works by blocking an enzyme encoded by the BRAF gene. The enzyme is a kinase and like all kinases, its job is to add phosphate groups onto targets. The addition (or removal) of phosphate groups from proteins frequently acts like an on-off switch. This drug blocks the activity of a specific mutated form of BRAF. The presence of the mutation in the cancer must be identified by a special (FDA-approved) test before the drug can be used on a patient. It should be noted that Vemurafenib does not inhibit wild-type BRAF or other Raf kinases.1
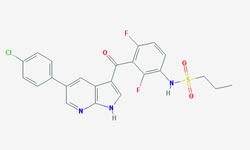
The diagram below shows the 3D molecular structure of Vemurafenib.
- 1 Chu, E., & DeVita, V. T. (2015). Physicians' cancer chemotherapy drug manual 2015. Burlington, MA: Jones & Bartlett Learning.
Vemurafenib can have some serious side effects, which include the development of cutaneous squamous cell cancers and keratoacanthomas as a result of treatment. Other, less severe toxicities include skin reactions, elevations in serum bilirubin and photosensitivity.1
- 1 Chu, E., & DeVita, V. T. (2015). Physicians' cancer chemotherapy drug manual 2015. Burlington, MA: Jones & Bartlett Learning.
