Tositumomab
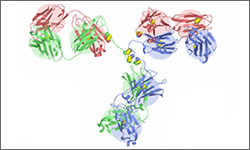
Tositumomab (Bexxar®) is a monoclonal antibody treatment targeted at the CD20 antigen. Tositumomab is used in the treatment of relapsed or refractory (treatment resistant) Non-Hodgkin's Lymphoma. Tositumomab is administered as 2 sets of infusions, a non-radioactive antibody and then a radioactive antibody. 1
- 1 Bexxar Prescribing Information. GlaxoSmithKline and Corixa Corporation. 2013. [https://www.gsksource.com/pharma/content/dam/GlaxoSmithKline/US/en/Prescribing_Information/Bexxar/pdf/BEXXAR.PDF]
This treatment is different from most antibody-based treatmets. In this case, the antibody has been permanently linked to a radioactive form of iodine (I131). The antibody is used to deliver the radioactive iodine to cancer cells which are then destroyed by the radiation. This form of treatment is termed radioimmunotherapy. Note that the image shown is a general structure of an antibody. There are four proteins bound together in a 'Y' shape. The two top tips of the Y are where the antibody binds with its target. This means that each antibody molecule can bind to two identical target regions.1
- 1 Chu, E., & DeVita, V. T. (2015). Physicians' cancer chemotherapy drug manual 2015. Burlington, MA: Jones & Bartlett Learning.
The most common side effect associated with tositumomab treatment is lowered blood cell count. Other common side effects are mostly infusion-related, and they include: weakness, fever, nausea, infection, cough. Severe allergic reactions may also occur.1
- 1 Chu, E., & DeVita, V. T. (2015). Physicians' cancer chemotherapy drug manual 2015. Burlington, MA: Jones & Bartlett Learning.
Patients should not become pregnant while undergoing tositumomab therapy and therapy should not be given before pregnancy is ruled out. Contraception should be used by males and females during and 12 months after treatment has ended. Patients who have already received murine proteins and may have mounted an anti-mouse immune response are at a higher risk for a reaction of this sort. In some patients hypothyroidism (reduced thyroid gland function) may occur. For this reason thyroid blocking agents must be administered with tositumomab. Treatment and screening for hypothyroidism should continue on an annual basis after treatment ends. Because this therapy includes a radioactive material that is inside the body for some period of time, patients should be given special instructions and precautions to prevent harm to any other person.1
- 1 Chu, E., & DeVita, V. T. (2015). Physicians' cancer chemotherapy drug manual 2015. Burlington, MA: Jones & Bartlett Learning.
