Tamoxifen
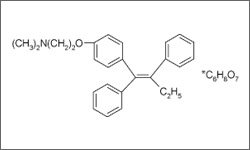
Tamoxifen is used to treat breast cancer in both pre- and postmenopausal women with advanced or metastatic breast cancer. It is also given to reduce the risk of invasive breast cancer in women who have been treated for ductal carcinoma in situ (DCIS). Tamoxifen is taken in tablet form. Patients that are taking tamoxifen to reduce the risk of invasive breast cancer, whether in a high risk population or who have a history of DCIS, will normally receive this treatment for a period of 5 years.1
- 1 Chu, E., & DeVita, V. T. (2015). Physicians' cancer chemotherapy drug manual 2015. Burlington, MA: Jones & Bartlett Learning.
Tamoxifen (Nolvadex®) is a molecule that competes with estrogen for binding sites on estrogen receptors in certain tissues of the body. This competition with estrogen limits cell growth in some tissues, as it leads to the inhibition of transcriptional and signaling processes that are required for cell growth.1
The diagram above shows the 3D structure of 4-Hydroxytamoxifen.
- 1 Chu, E., & DeVita, V. T. (2015). Physicians' cancer chemotherapy drug manual 2015. Burlington, MA: Jones & Bartlett Learning.
Common side effects include a variety of menopausal symptoms: hot flashes, vaginal discharge or bleeding, menstrual irregularities. Other side effects include: fluid retention, headache, lethargy, skin rash and myelosuppression.1
- 1 Chu, E., & DeVita, V. T. (2015). Physicians' cancer chemotherapy drug manual 2015. Burlington, MA: Jones & Bartlett Learning.
Patients being treated with tamoxifen are also at increased risk for blood clots in the legs and lungs, and also for strokes. These events are serious but are rare. Tamoxifen may also increase the risk of endometrial cancer in patients receiving treatment that have not undergone a hysterectomy. Patients should not become pregnant while taking tamoxifen or within 2 months after treatment is stopped as the drug could harm the unborn child. Also, oral contraceptives should not be used while taking tamoxifen. Therefore other forms of birth control must be used, such as condom or diaphragm.1, 2
- 1 Nolvadex.. Prescribing Information. AstraZeneca. May, 2002. [http://www.nolvadex.com]
- 2 Chu, E., & DeVita, V. T. (2015). Physicians' cancer chemotherapy drug manual 2015. Burlington, MA: Jones & Bartlett Learning.
