Rolapitant
Brand name:
Varubi®
IUPAC:
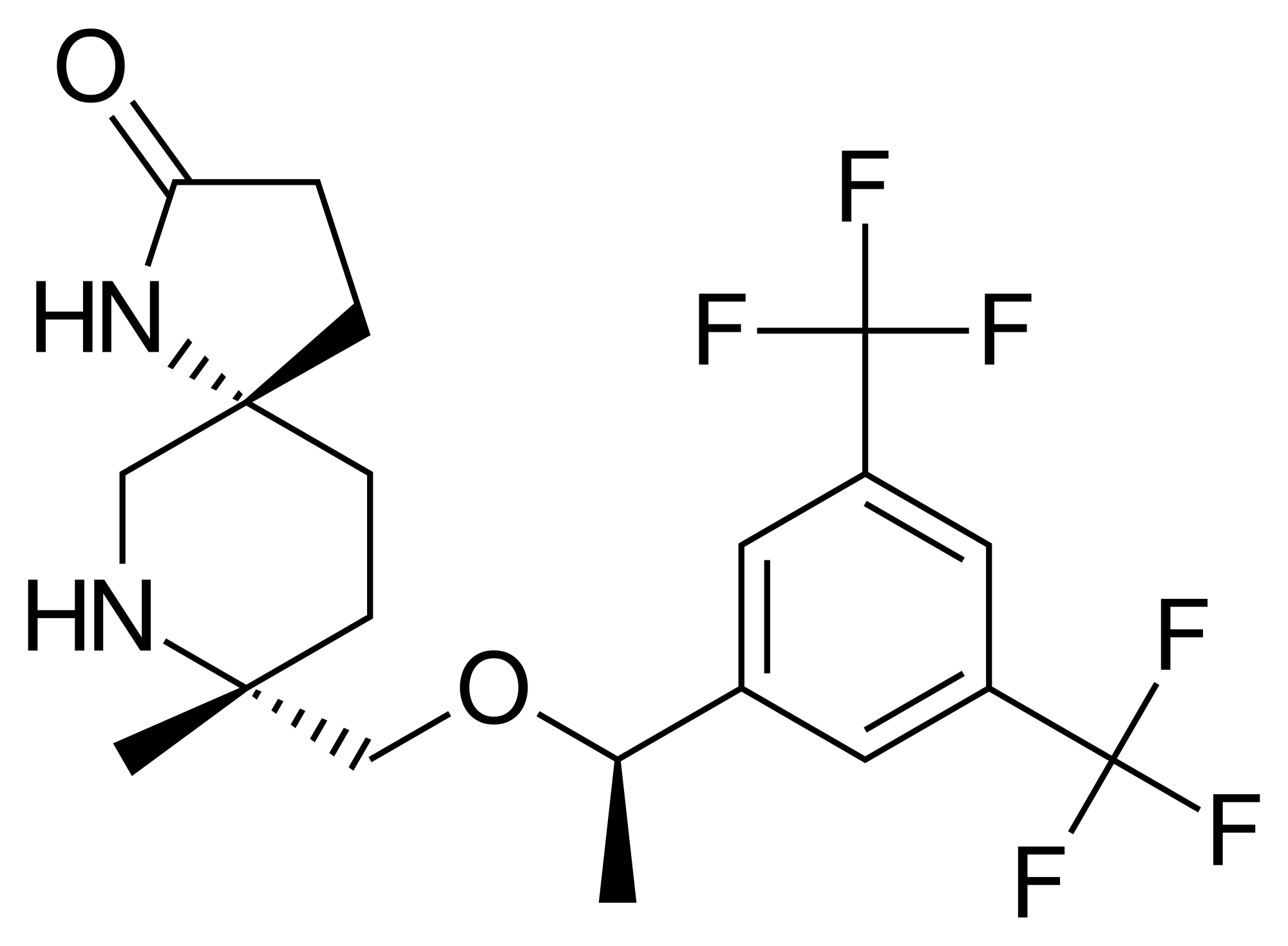
(5S,8S)-8-[[(1R)-1-[3,5-bis(trifluoromethyl)phenyl]ethoxy]methyl]-8-phenyl-1,9-diazaspiro[4.5]decan-2-one
FDA approval:
Yes
Mechanism:
Rolapitant (Varubi®) is a human substance P/neurokinin-1 (NK-1) receptor antagonist. In other words, rolapitant prevents the binding of human substance P/neurokinin-1 (NK-1) receptor with its agonist, inhibiting the activation of the receptors. Activation of NK-1 receptors is associated with the nausea and vomiting induced by certain chemotherapy drugs. Rolapitant works by blocking the actions of neurokinin and substance P, which are natural substances in the brain that produce nausea and vomiting.1
The diagram below shows the 3D molecular structure of Rolapitant.
- 1 Rolapitant. MedlinePlus. 2016. https://medlineplus.gov/druginfo/meds/a615041.html
