Panitumumab
Vectibix is classified as a monoclonal antibody and signal transduction inhibitor by binding to epidermal growth factor receptors (EGFR). Vectibix is used to treat colorectal cancer that has spread. It is used to treat colon cancers that express EGFR and disease that has gotten worse either on or following fluoropyrimidine, oxaliplatin and irinotecan containing chemotherapy regimens.
Vectibix is injected into a vein through an IV. A healthcare provider will give you this injection. Before you receive this medicine, you may need to undergo a biopsy to make sure panitumumab is the right medication to treat your cancer. Vectibix must be given slowly, and the IV infusion can take up to 90 minutes to complete. Vectibix is usually given once every 2 weeks. Follow your doctor's instructions. While using panitumumab, you may need frequent blood tests. Your skin and vision may also need to be checked. Panitumumab can have long lasting effects on your body. You may need frequent medical tests for up to 8 weeks after you stop using this medicine.
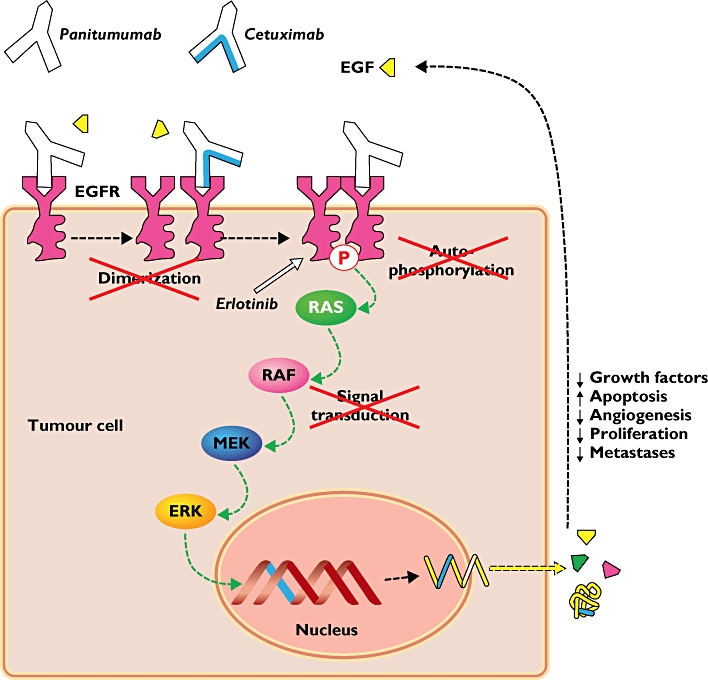
The EGFR is a transmembrane glycoprotein that is a member of a subfamily of type I receptor tyrosine kinases, including EGFR, HER2, HER3, and HER4. EGFR is constitutively expressed in normal epithelial tissues, including the skin and hair follicle. EGFR is overexpressed in certain human cancers, including colon and rectum cancers. Interaction of EGFR with its normal ligands (eg, EGF, transforming growth factor-alpha) leads to phosphorylation and activation of a series of intracellular proteins, which in turn regulate transcription of genes involved with cellular growth and survival, motility, and proliferation. Signal transduction through the EGFR results in activation of the wild-type KRAS protein. However, in cells with activating KRAS somatic mutations, the KRAS-mutant protein is continuously active and appears independent of EGFR regulation.
Panitumumab binds specifically to EGFR on both normal and tumor cells, and competitively inhibits the binding of ligands for EGFR. Nonclinical studies show that binding of panitumumab to the EGFR prevents ligand-induced receptor autophosphorylation and activation of receptor-associated kinases, resulting in inhibition of cell growth, induction of apoptosis, decreased proinflammatory cytokine and vascular growth factor production, and internalization of the EGFR. In vitro assays and in vivo animal studies demonstrate that panitumumab inhibits the growth and survival of selected human tumor cell lines expressing EGFR.
The diagram below shows the 3D molecular structure of Panitumumab in complex with epidermal growth factor receptor domain 3.
Some people receiving a Vectibix injection have had a reaction to the infusion (when the medicine is injected into the vein). Tell your caregiver right away if you feel dizzy, nauseated, light-headed, or have a fever, chills, chest tightness, or trouble breathing during the injection. Common Vectibix side effects may include: acne, dry skin, rash, itching; swelling or irritation around your fingernails or toenails; loss of appetite, nausea, diarrhea; blisters or ulcers in your mouth, red or swollen gums, trouble swallowing; tired feeling, weakness; or low potassium - confusion, uneven heart rate, extreme thirst, increased urination, leg discomfort, muscle weakness or limp feeling.
Serious effects that can result from panitumumab treatment include dermatologic toxicity including dermatitis acneiform, redness, paronychia (infection at the base of the fingernail or toenail), rash, skin exfoliation, dry skin and skin fissures; infusion reactions (anaphylactic shock, bronchospasm, fever, chills and low blood pressure); and pulmonary fibrosis. Due to a potential harm to the developing fetus, women receiving this treatment should take contraceptive measures and pregnant women should be warned of this risk. New mothers should not nurse infants during or immediately following this treatment regimen. Panitumumab is not recommended for use in combination chemotherapy regimens. Since sunlight can enhance skin reactions due to panitumumab, patients should limit sun exposure during treatment.1
- 1 Chu, E., & DeVita, V. T. (2015). Physicians' cancer chemotherapy drug manual 2015. Burlington, MA: Jones & Bartlett Learning.
