Paclitaxel
In December of 1992, the FDA approved paclitaxel for the treatment of ovarian cancer. Today the drug is used for a several types of cancer, including ovarian, breast, small cell and large cell lung cancers, and Kaposi's sarcoma. Paclitaxel is administered as a series of intravenous injections.1
- 1 Chu, E., & DeVita, V. T. (2015). Physicians' cancer chemotherapy drug manual 2015. Burlington, MA: Jones & Bartlett Learning.
Paclitaxel (Taxol®) binds to microtubules and prevents their breakdown. The movement of the replicated chromosomes during mitosis requires both polymerization of tubulin to form microtubules as well as the breakdown of those microtubules. In the presence of paclitaxel, chromosomes are unable to move to opposite sides of the dividing cell because microtubules are not broken down. Cell division is halted, and cell death is induced.1
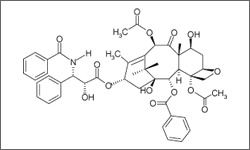
The diagram below show the 3D molecular structure of Paclitaxel.
- 1 Chu, E., & DeVita, V. T. (2015). Physicians' cancer chemotherapy drug manual 2015. Burlington, MA: Jones & Bartlett Learning.
Common side effects include reduction in bone marrow function which may result in anemia, blood in stools or black stools, fast or irregular heartbeat, fever, chills, lower back pain, numbness or tingling of the hands or feet, mouth sores, painful, bloody, or difficult urination, swelling of the face, lips, or throat, nausea, vomiting, or diarrhea, unusual bleeding or bruising, wheezing or trouble breathing, hair loss.1
- 1 Chu, E., & DeVita, V. T. (2015). Physicians' cancer chemotherapy drug manual 2015. Burlington, MA: Jones & Bartlett Learning.
The drug may cause birth defects. Patients should not take this drug if they are pregnant or plan to become pregnant. Breast feeding should also be avoided by patients taking paclitaxel. Immunosuppression may lead to increased risk of infection. Patients should tell their clinician if they are currently taking any other medications.1
- 1 Chu, E., & DeVita, V. T. (2015). Physicians' cancer chemotherapy drug manual 2015. Burlington, MA: Jones & Bartlett Learning.
