Mitoxantrone
Malignancies for which mitoxantrone is used include: advanced, hormone-refractory Prostate cancer and Acute nonlymphocytic leukemia (ANLL). Mitoxantrone is also used in treatment of breast cancer and Non-Hodgkin's lymphoma. Also worth noting is that Mitoxantrone is administered as an intravenous infusion.1
- 1 Chu, E., & DeVita, V. T. (2015). Physicians' cancer chemotherapy drug manual 2015. Burlington, MA: Jones & Bartlett Learning.
Mitoxantrone (Novantrone®) is structurally similar to doxorubicin, as it is an antitumor antibiotic. It exerts its effects on cancer cells primarily through two mechanisms: Mitoxantrone wedges between the bases of DNA (intercalation) and blocks DNA synthesis and transcription, causing cross links and strand breaks. Mitoxantrone inhibits the activity of an enzyme, topoisomerase type II. This leads to breaks in the genomic DNA.1
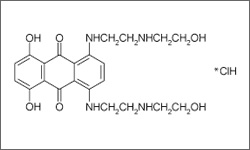
The diagram below shows the 3D molecular structure of Mitoxantrone.
- 1 Chu, E., & DeVita, V. T. (2015). Physicians' cancer chemotherapy drug manual 2015. Burlington, MA: Jones & Bartlett Learning.
Patients should note that after an infusion the urine may have a temporary blue-green tinting. Mitoxantrone is a suppressor of bone marrow activity. It is important to monitor blood cell and platelet counts throughout the duration of treatment with blood tests before each infusion. Mitoxantrone may also have toxic effects the liver, which require liver function to be monitored routinely. Also, heart function must be monitored to avoid irreversible effects of toxicity. Evaluations of the function of these systems should be made before each infusion. Mitoxantrone should not be taken by women who are pregnant and patients should not become pregnant while using this drug, as it may have harmful affects on the developing fetus. It is unclear whether this drug may have effects on fertility after treatment has ended.1
- 1 Chu, E., & DeVita, V. T. (2015). Physicians' cancer chemotherapy drug manual 2015. Burlington, MA: Jones & Bartlett Learning.
