Imatinib Mesylate
Imatinib was designed to treat chronic myeloid leukemia (CML). In CML, a chromosomal rearrangement in white blood cells fuses two genes together. Imatinib is also used in the treatment MDS/MPD, which are associated with rearrangement of the PDGFR gene. It should be noted that Imatinib has a vast range of different doses, so you should talk to your doctor to figure out the best dose for you. Imatinib is taken through an oral tablet.1
- 1 Chu, E., & DeVita, V. T. (2015). Physicians' cancer chemotherapy drug manual 2015. Burlington, MA: Jones & Bartlett Learning.
Imatinib mesylate (Gleevec®) is a tyrosine kinase inhibitor that works to prevent the growth-inducing signals sent by enzymes found in some cancerous cells. The product of the BCR-ABL gene is a kinase that adds phosphate groups onto target molecules (substrates). Imatinib binds to the the ATP binding site of the enzyme. If it is unable to bind ATP, the BCR-ABL protein cannot carry out its kinase activity. The signals sent by the enzyme would act to stimulate cell division so Imatinib inhibits tumor growth of cancerous cells.1
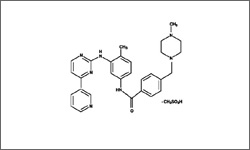
The diagram above shows the 3D molecular structure of Imatinib Mesylate.
- 1 Chu, E., & DeVita, V. T. (2015). Physicians' cancer chemotherapy drug manual 2015. Burlington, MA: Jones & Bartlett Learning.
Common side effects include frequent nausea and vomiting, diarrhea, leg aches or cramps, swelling of the face, altered bone marrow function and fluid retention. These side effects are usually mild or moderate and frequently occur during the first month of treatment and may get better after initial treatments.1
- 1 Chu, E., & DeVita, V. T. (2015). Physicians' cancer chemotherapy drug manual 2015. Burlington, MA: Jones & Bartlett Learning.
