Doxorubicin
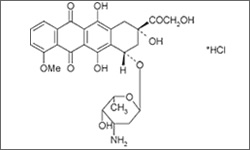
Doxorubicin is useful in a wide range of cancers, including: breast cancer, lymphoma, soft tissue sarcoma, ovarian cancer and many more. There are only a few cancer types are unresponsive to the drug, which include: colon cancer, melanoma, chronic leukemias and renal cancer. Doxorubicin is administered intravenously. In a newer formulation, Doxil, the drug is surrounded by a lipid 'bubble' (membrane). This formulation is an attempt to reduce the toxicity of the drug by blocking its effects on normal tissues.1
- 1 Chu, E., & DeVita, V. T. (2015). Physicians' cancer chemotherapy drug manual 2015. Burlington, MA: Jones & Bartlett Learning.
Doxorubicin is an anthracycline antibiotic that exerts its effects on cancer cells via two different mechanisms. It acts as an intercalating agent and wedges between the DNA bases thus blocking DNA synthesis and transcription. The drug also inhibits the activity of an enzyme, topoisomerase type II. This leads to breaks in the genomic DNA. Both of these mechanisms result in DNA disruption that ultimately can lead to the death of the cell.1 Interestingly, several studies have reported that green tea enhances the antitumor activity of doxorubicin.2
Below is the structure of a 3D Doxorubicin conformer.
- 1 Chu, E., & DeVita, V. T. (2015). Physicians' cancer chemotherapy drug manual 2015. Burlington, MA: Jones & Bartlett Learning.
- 2 Cheng, T., Liu, J., Ren, J., Huang, F., Ou, H., Ding, Y., … Shi, L. (2016). Green Tea Catechin-Based Complex Micelles Combined with Doxorubicin to Overcome Cardiotoxicity and Multidrug Resistance. Theranostics, 6(9), 1277–1292. http://doi.org/10.7150/thno.15133
Common side effects include: decreased blood cell counts, increased risk of infection and bleeding, loss of appetite, stomatitis, alopecia (hair loss), Nausea and vomiting, mouth sores, Birth defects, liver toxicity, and acute arrhythmia. Cardiac toxicity becomes relevant at high doses. If present, the cardiomyopathy may lead to irreversible congestive heart failure.1 Due to this concern, patients should talk to their doctors about any cardiac conditions or complications. Slow intravenous infusion can reduce cardiac toxicity (by lowering blood concentrations of the drug). However, this method can be problematic because it lasts for 48-96 hours.2
- 1 Chu, E., & DeVita, V. T. (2015). Physicians' cancer chemotherapy drug manual 2015. Burlington, MA: Jones & Bartlett Learning.
- 2 Rygiel, K. (2016). Benefits of antihypertensive medications for anthracycline- and trastuzumab-induced cardiotoxicity in patients with breast cancer: Insights from recent clinical trials. Indian Journal of Pharmacology, 48(5), 490–497. http://doi.org/10.4103/0253-7613.190719
