Bevacizumab
Bevacizumab is used as in combination with chemotherapy for the treatment of metastatic colorectal cancer. Bevacisumab was additionally approved in 2006 in combination with carboplatin and paclitaxel for the treatment of non-squamous, non-small cell lung cancer that is unresectable, advanced, recurrent or metastatic.1 Bevacizumab is also approved for use in non-small cell lung cancer, glioblastoma, renal cell cancer and cervical cancer.2
- 1 Cohen, M., Gootenberg, J., Keegan, P., & Pazdur, R. (2007). FDA drug approval summary: bevacizumab (Avastin) plus Carboplatin and Paclitaxel as first-line treatment of advanced/metastatic recurrent nonsquamous non-small cell lung cancer. The Oncologist, 12(6), 713–8. (Original work published June 2007) [PUBMED]
- 2 Chu, E., & DeVita, V. T. (2015). Physicians' cancer chemotherapy drug manual 2015. Burlington, MA: Jones & Bartlett Learning.
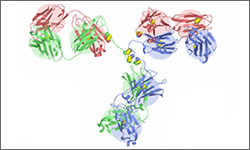
Bevacizumab (Avastin®) is a monoclonal antibody treatment that attacks the VEGF (vascular endothelial growth factor) through binding and prevents it from binding to its receptors on endothelial cells. This interrupts the signal for necessary blood vessel growth in a tumor. Note that the image shown is a general structure of an antibody, which in turn effects the process of VEGFR-signaling. There are four proteins bound together in a 'Y' shape. The two top tips of the Y are where the antibody binds with its target. This means that each antibody molecule can bind to two identical target regions.1
- 1 Chu, E., & DeVita, V. T. (2015). Physicians' cancer chemotherapy drug manual 2015. Burlington, MA: Jones & Bartlett Learning.
Serious side effects include gastrointestinal abscesses, perforations, and bleeding. Treatment with this drug may also delay wound healing, which includes delayed healing of wounds from surgical procedures.1 Less severe side effects include: weakness, diarrhea, low white blood cell count, abdominal pain, headache, hypertension.2
