Bendamustine
The FDA has approved bendamustine for the treatment of chronic lymphocytic leukemia (CLL). Treanda® is the first drug approved for CLL treatment since 2001.
Bendamustine forms organic ions that bind to other ions to induce cell death in both latent and dividing cells. The exact mechanism by which these linkages cause cell death is still unknown, however Bendamustine is known to form cross-links with DNA, which causes breakage of strands and inhibition of DNA synthesis.1
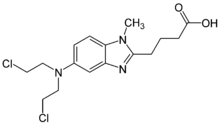
The diagram below shows the 3D molecular structure of Bendamustine.
- 1 Chu, E., & DeVita, V. T. (2015). Physicians' cancer chemotherapy drug manual 2015. Burlington, MA: Jones & Bartlett Learning.
Side Effects include: Hypersensitivity reactions (including rash, facial swelling or difficulty, breathing), Fatigue, Nausea and vomiting, Diarrhea, Rash or itching.1
- 1 Chu, E., & DeVita, V. T. (2015). Physicians' cancer chemotherapy drug manual 2015. Burlington, MA: Jones & Bartlett Learning.
Potential serious adverse reactions include myelosuppression (low levels of platelets, red blood cells and white blood cells), infection, infusion reactions, tumor lysis syndrome and skin reactions. Bendamustine can cause harm to a fetus during development, therefore pregnant women should be warned of this risk and women should avoid becoming pregnant during treatment and for 3 months afterward. Likewise, men receiving bendamustine treatment should use contraceptive measures during and for 3 months after treatment. New mothers should not nurse infants during treatment as it is possible for the drug to be present in breast milk. Bendamustine treatment is not recommended for patients with moderate or severe kidney or liver impairment. Bendamustine should also not be used in patients with a known allergy to bendamustine or mannitol. Caution should be used in the treatment of patients also receiving CYP1A2 inhibitors or inducers since these drugs have the potential to alter the blood levels of bendamustine.
