Azacitidine
Vidaza® (Azacitidine) was approved by the FDA in 2004. It is an orphan product, which means it was developed to treat rare diseases and conditions that affect less than 200,000 people in the United States. Azacitidine is used to treat patients with the following FAB myelodysplastic syndrome (MDS) subtypes: refractory anemia (RA), refractory anemia with ringed sideroblasts (RARS), refractory anemia with excess blasts (RAEB), refractory anemia with excess blasts in transformation (RAEB-T), and chronic myelomonocytic leukemia (CMMoL). It is injected intravenously (IV), often 7 days in a row, every 4 weeks, for 4 treatment cycles or more.
Azacitidine is a DNA demethylating agent. Methylation of DNA is linked to decreased activity of the targeted region. In cancer, genes that control cell division can be turned off by methylation. Azacitidine can reverse the methylation, allowing the genes to function again, ultimately slowing cell growth. Azacitidine is also an antimetabolite. Antimetabolites are chemicals that look like things normally used in cells. They can interact with proteins and other cell components, but these interactions don't give the same results as the normally used item. Because of this activity, azacitidine can directly cause the death of cancer cells.
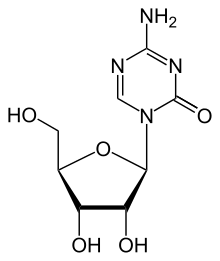
The diagram above shows the 3D molecular structure of Azacitidine.
