Mechlorethamine topical
Apply immediately or within 30 minutes after removal from the refrigerator. Return VALCHLOR to the refrigerator immediately after each use.
Apply to completely dry skin at least 4 hours before or 30 minutes after showering or washing. Allow treated areas to dry for 5 to 10 minutes after application before covering with clothing.
Emollients (moisturizers) may be applied to the treated areas 2 hours before or 2 hours after application.
Do not use occlusive dressings on areas of the skin where VALCHLOR was applied.
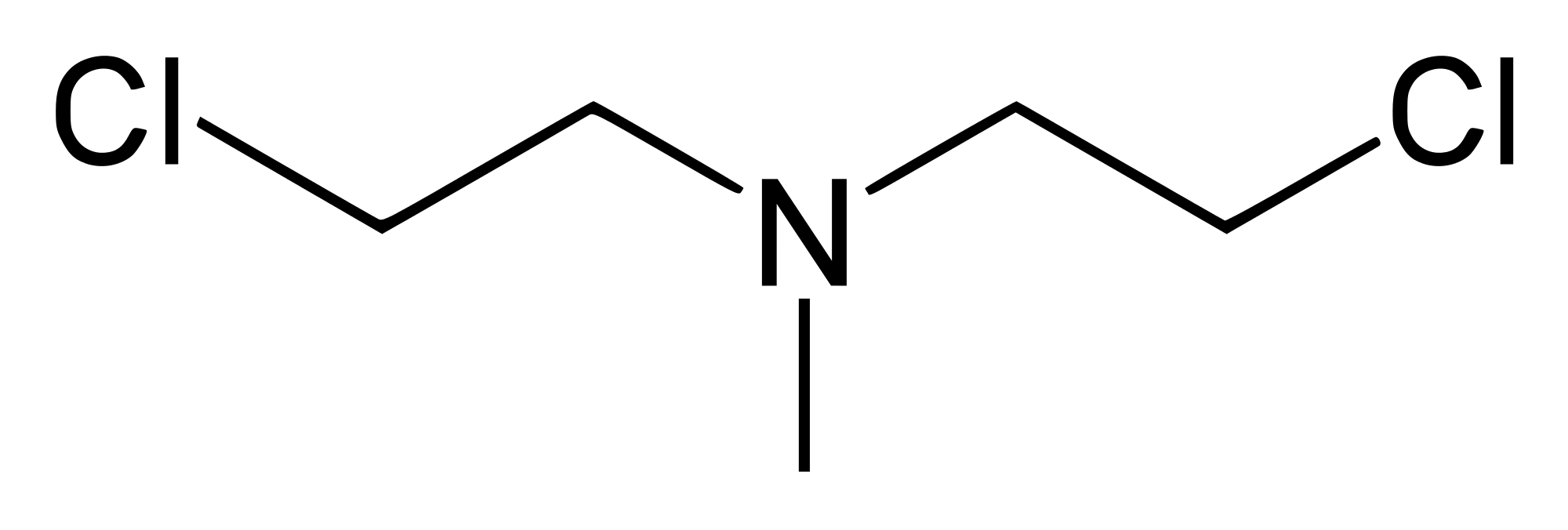
Valchlor is a gel formulation of mechlorethamine, an alkylating agent which inhibits rapidly proliferating cells. Valchlor is specifically indicated for the topical treatment of Stage IA and IB mycosisfungoides-type cutaneous T-cell lymphoma in patients who have received prior skin-directed therapy. Valchlor is supplied as a gel for topical administration. The recommendation is to apply a thin film of Valchlor gel once daily to affected areas of the skin. Upon improvement, treatment with Valchlor can be restarted at a reduced frequency of once every 3 days. If reintroduction of treatment is tolerated for at least one week, the frequency of application can be increased to every other day for at least one week and then to once daily application if tolerated.
Valchlor is a gel formulation of mechlorethamine, also known as nitrogen mustard, an alkylating agent which inhibits rapidly proliferating cells.
Side effects of Valchlor include: dermatitis, pruritus, bacterial skin infection, skin ulceration or blistering, and hyperpigmentation.
