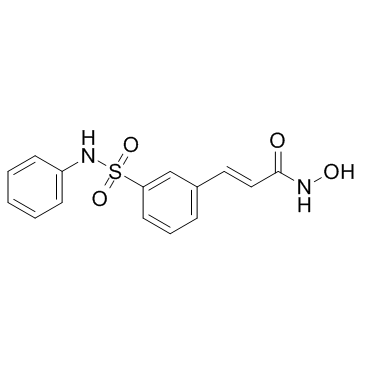
Belinostat
Beleodaq is injected into a vein through an IV. A healthcare provider will give you this injection. The injection must be administered slowly, and the IV infusion can take at least 30 minutes to complete. Beleodaq is given in a 21-day treatment cycle, and you may only receive the medicine during the first 5 days of each cycle. Your doctor will determine how long your treatment will last. You may be given medication to prevent nausea, vomiting, or diarrhea while you are receiving belinostat. You will need frequent medical tests to be sure this medicine is not causing harmful effects. Your blood will need to be tested every week during each treatment cycle. Your cancer treatments may be delayed based on the results of these tests. Beleodaq can lower blood cells that help your body fight infections and help your blood to clot. This can make it easier for you to bleed from an injury or get sick from being around others who are ill.
The U.S. Food and Drug Administration approved Beleodaq (belinostat) for the treatment of patients with peripheral T-cell lymphoma (PTCL), a rare and fast-growing type of non-Hodgkin lymphoma (NHL). The action was taken under the agency’s accelerated approval program.PTCL comprises a diverse group of rare diseases in which lymph nodes become cancerous. In 2014, the National Cancer Institute estimates that 70,800 Americans will be diagnosed with NHL and 18,990 will die. PTCL represents about 10 to 15 percent of NHLs in North America. Beleodaq works by stopping enzymes that contribute to T-cells, a type of immune cell, becoming cancerous. It is intended for patients whose disease returned after treatment (relapsed) or did not respond to previous treatment (refractory).
Side effects of Beleodaq include: nausea, fatigue, fever (pyrexia), low red blood cells (anemia), and vomiting.
