Nilotinib
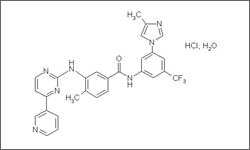
Nilotinib is approved to treat chronic or accelerated phase Philadelphia chromosome-positive chronic myelogenous leukemia (CML) that is resistant to prior therapy with imatinib.1
- 1Chu, E., & DeVita, V. T. (2015). Physicians' cancer chemotherapy drug manual 2015. Burlington, MA: Jones & Bartlett Learning.
Nilotinib (Tasigna®) is a kinase inhibitor that binds to the inactive form of the Abl protein to inhibit Bcr-Abl kinase activity.1
The diagram below shows the 3D molecular structure of Nilotinib.
- 1Chu, E., & DeVita, V. T. (2015). Physicians' cancer chemotherapy drug manual 2015. Burlington, MA: Jones & Bartlett Learning.
The most common toxicities produced by nilotinib are myelosuppression (low blood counts), QT prolongation (increased duration of cardiac ventricular repolarization), elevated serum lipase, liver toxicity and electrolyte abnormalities (irregular levels of phosphate, potassium, calcium and sodium). Nilotinib is not recommended for patients with low potassium levels, low magnesium levels or long QT syndrome. Additionally, nilotinib contains lactose so patients with galactose intolerance, severe lactase deficiency or problems with glucose-galactose absorption should be treated with caution. Due to potential harm to the fetus, prospective mothers should be warned before beginning treatment and women should avoid becoming pregnant while receiving nilotinib. Mothers should not breast feed infants while undergoing nilotinib treatment. Due to the possibility of drug interaction, administration of nilotinib along with CYP3A4 inhibitors or drugs that prolong QT interval is not advised.1
- 1http://www.us.tasigna.com/patient/ph-cml-treatment.jsp
