Humans aren't the only ones affected by cancer. This section contains information detailing cancer in domesticated animals. If the animal/cancer type in which you are interested is not yet presented, please check back because we add new information regularl. Further information on the topics on this page can also be found in most introductory biology textbooks, we recommend the textbook, Campbell Biology, 11th edition.1
Cancer in Pets
According to the American Veterinary Medical Association (AVMA), cancer causes almost 50% of deaths in pets over the age of 10. Some common types of cancers in pets include: skin, breast, head and neck, lymphoma, leukemia, testicular, abdominal, and bone. Examples of cancers commonly found in pets that are also commonly found in humans are lymphoma, melanoma, and osteosarcoma. 2 3
Of the two most common household pets, dogs tend to get cancer at a higher rate than cats. The cancer may be treatable depending on cancer type. Cancer treatment options for dogs and cats are similar to those for humans. Surgery, radiation therapy, chemotherapy, and immunotherapy are all considered and will usually result in less side effects in animals than in humans. Veterinarians will tailor treatment dosages to optimize efficacy and minimize side effects. 3
Table of Contents:
Cancer in Cats
Cats are susceptible to a variety of cancers. Among the most common are lymphoma, squamous cell carcinoma (skin cancer), mammary cancer, mast cell tumors, oral tumors, fibrosarcoma (soft tissue cancer) , osteosarcoma (bone cancer), respiratory carcinoma, intestinal adenocarcinoma, and pancreatic/liver adenocarcinoma. The disease has become so prevalent that it is now the most common cause of death in cats.4
Certain breeds are more prone to certain cancers than others. Signs and symptoms differ depending on the type and stage of the cancer. Detection and diagnosis requires some detective work. Tumors that are visible and/or detectable by touch are most easily identified. Vets often perform additional tests to make an accurate diagnosis. Along with a physical exam, they may perform blood and urine tests, cytology, imaging and biopsies.
Treatment options vary and include surgery, chemotherapy, radiation, immunotherapy, photodynamic therapy, or a combination of these. In many cases, cancer can be successfully treated. Early detection and diagnosis is critical. Regular visits to the veterinarian can help prevent and manage cancer. The American Veterinary Medical Association recommends twice-a-year wellness exams for all cats.5 Because the causes of cancer in cats are similar to those in humans, risk can be reduced by lowering the animal's exposure to harmful carcinogens, including tobacco smoke.6
Feline Leukemia and the Feline Leukemia Virus
Overview and Transmission
In humans, some viruses can lead to cancer. An example is human papilloma virus (HPV) which is the causative agent of most cases of cervical cancer. Viral infection can also lead to cancer in animals. Feline leukemia (FeLV; informally known as "Fee-Leuk") is an RNA virus (a retrovirus) that infects less than 2% of healthy, domestic cats in the U.S.7 8 Infection is more prevalent in high-risk populations (i.e. cats with outdoor access and/or frequent social interactions).7
The virus is spread from one cat to another via saliva, nasal secretions, feces, and milk.9 10 It is transmitted during various forms of contact, from friendly grooming to not-so-friendly biting. The virus can also be passed to a developing kitten during pregnancy. The age and time of infection affect the progression and clinical outcome of the virus. Kittens are more likely to be infected and more likely to develop more severe complications.11 In most cases, FeLV initially infects lymphocytes in the back of the throat (oropharynx), which travel to the bone marrow, where virally infected cells are produced very rapidly.12
In most environments, the Feline leukemia virus cannot survive for long outside of the host. It can be 'killed' (made harmless) with soap and disinfectants. At this time, studies show no evidence that FeLV can be transmitted from infected cats to humans.13 However, because FeLV positive cats may carry other diseases, infants, elderly individuals, and immunosuppressed individuals may want to avoid contact.
Symptoms
Infected animals may develop anemia, lymphoma, and other conditions. The FeLV-C subtype binds to and impedes the function of a heme transport protein on the surface of developing red blood cells. The result is a decrease in red blood cell numbers (anemia).14 15 Signs of anemia in cats include paleness of the skin, tongue, gums, and mucous membranes surrounding the eye. FeLV- induced lymphomas are some of the most frequent tumors seen in cats. Symptoms depend on the location of the tumor, and may include weight loss, rough hair coat, loss of appetite, vomiting, diarrhea, respiratory distress, swelling of the lymph nodes and more. FeLV is also linked to diseases of the kidneys, joints, lymph nodes, small intestine, liver and nervous system. Depression of the immune system makes infected cats more susceptible to infections. As a result, they may be infected by organisms that healthy cats usually fend off. It is possible for FeLV positive cats to remain healthy. However, the prognosis is poor for cats with persistent active infection.12
Detection of FeLV
Veterinarians use several different laboratory tests to detect FeLV. These include 1) an antigen enzyme-linked immunosorbent assay (ELISA) and 2) an indirect immunofluorescent antibody assay (IFA). Both tests use blood samples to detect the presence of a protein that indicates FeLV infection. This protein is called p27, and is part of the structure of the virus.12 Inconclusive results may require additional testing with other methods, such as a specific type of polymerase chain reaction (PCR), which can detect FeLV DNA in infected animals.
Treatment for FeLV Infection
Currently there is no cure for FeLV. However there are a number of things that pet owners can do to manage FeLV positive cats, as recommended by veterinarians and experts:
- Visit the vet every 6 months for a physical examination, complete blood count, urine analysis, and other tests
- Carefully monitor the cat's health (i.e. measure and record weight loss)
- Provide cat(s) with a healthy and balanced diet. Avoid uncooked meat and eggs, as well as unpasteurized dairy products to prevent food-borne infections
- Confine cats to indoor environments to reduce exposure to harmful organisms
Antiviral therapy has been used in FeLV positive cats, but the effectiveness of the drugs is limited and they can be toxic. Medications to control and restore the immune system (immunomodulators) are also used. However data to support the proposed health benefits of these agents are limited.11 16
Prevention of FeLV Infection
Infection with FeLV can be prevented by vaccination. The vaccine is classified as non-core, which means it can be considered optional. However both the American Association of Feline Practitioners (AAFP) and the European Advisory Board on Cat Disease (ABCD) recommend that all cats with uncertain FeLV status and/or are at risk of exposure be vaccinated. Kittens are often vaccinated at 8-9 weeks of age and again at 12 weeks of age. Research shows that the vaccine will confer immunity for up to 1 year. Many vets recommend a booster vaccination 1 year after initial vaccination and annually thereafter. Because cats become less susceptible with age, some vets consider vaccination every 2-3 years sufficient for older animals.12
Visit the following sections to learn more about:
Feline Squamous Cell Carcinoma
Introduction
Squamous Cell Carcinoma (SCC) is a cancer that occurs in cats and dogs. The tumors can appear many places but are seen most commonly on and around the eyes, ears, nose, mouth, and areas with little hair.
Risk Factors/Detection/Staging
The primary cause of SCC is sunlight over-exposure, which is especially harmful to fair-haired cats. Age and exposure to cigarette smoke also increase the risk for SCC.17
At first, SCC tumors look much like other common skin irritations, making them difficult to identify. Scabs, hair loss, irritated skin, loss of teeth with limited healing, and raised red bumps on the skin are all possible sign of SCC.17
Crusty sores can appear and develop into deep ulcers that bleed when irritated. In later stages, the cancer may spread to the lymph nodes and lungs. Staging of the cancer may involve chest X-rays and testing lymph fluid.18
In advanced cases, SCC tumors can cause tissue death and destroy bone structure, leading to pain, discomfort, and possibly death.
Treatment
Several treatment options exist for SCC and they may be combined to increase success. As with other cancers, the earlier SCC is detected, the more likely the treatment will be successful. Tumors in the lower jaw or upper ear can be surgically removed. Vets attempt to preserve as much healthy tissue as possible. For tumors in the lower jaw, vets can perform a surgical Mandibulectomy, in which they attempt to remove the cancerous area(s). The amount of the jaw tissue and bone removed depends on the tumor. Possible side effects include difficulty eating and grooming, drooling, and poor appetite.17
Despite a cancer reoccurrence rate of 38% and post-operation complications, one survey revealed that over 80% of owners whose cats had surgery would choose this treatment again.17
Chemotherapy alone has not proven effective, but there is evidence that it may be effective when combined with other treatments. A combination of the chemotherapy agent mitoXANTRONE with external beam radiation therapy gave better results than either treatment option used alone.19
If the tumor is in the mouth, radiation should be considered with caution as it can cause severe inflammation. Side effects may prevent normal eating and require the use of a feeding tube.
Cryotherapy or freeze-surgery involves insertion of a probe that freezes the tumor, killing the cancer cells. Cryotherapy is an option for tumors under 1 cm (about 0.4 inch) in diameter. The rate of reoccurrence is high for larger tumors.20 Cryotherapy is a good option for tumors on the upper ear because the position allows for direct and aggressive treatment.
Photodynamic therapy (PT) is a newer treatment option for Squamous Cell Carcinoma, and is not available everywhere. This type of treatment involves putting a light-sensitive material in the tumor and exposing the tumor to a specific wavelength of light. This causes the light-sensitive chemical to become active and destroy the tumor cells.21 It can be used repeatedly and with other treatment options.22 The side effects include swelling, redness, sensitivity to light, and death of healthy tissue around the tumor site. A recent study followed 12 cats with SCC that were treated with PT. Researchers found that when used alone, PT resulted in shrinkage of shallow tumors of the nose and ear, but had little effect on deeper tumors. When used in combination with surgery, two cats exhibited a partial response to therapy, and one exhibited a complete response.21
Feline Mammary Cancer

Introduction
Feline mammary cancer (FMC) is the third most common cancer in female cats. It is very uncommon in males, but cases have been reported.23
The mammary gland tumors that result from FMC can be benign or malignant. However, research shows that 80-96% of mammary tumors are malignant, meaning that they can invade nearby tissues, and are capable of metastasis (spreading to distant parts of the body).23
24
Unfortunately malignant mammary tumors tend to be much more harmful.
Cats have four mammary glands on each side of their body, any of which can be affected by FMC. They are referred to as axillary, thoracic, abdominal, and inguinal glands. These glands are the sites of primary tumor growth. As with breast cancer in women, FMC is highly metastatic. As a result, this type of cancer can spread to the lungs, the lining of the lungs and chest cavity (pleura), liver, regional lymph nodes, and other parts of the body.
Genes that have been targeted in human breast cancer research have also been studied in feline mammary cancer. HER-2/neu is the gene that encodes the human epidermal growth factor receptor (type 2) protein. This protein resides on cell surfaces, where it can interact with growth signals. If gene amplification creates extra copies of the HER-2 gene, then its corresponding protein will also be overexpressed. This is thought to lead to increased cell proliferation. HER-2 overexpression has been detected in up to 30% of human breast cancer cases.23 Similarly, it occurs in 30% of feline mammary cancer cases.4
Risk Factors/Detection/Staging
While all feline breeds may be affected by FMC, some are at a higher risk than others.4
Studies have shown the Siamese breed to have twice the risk of developing this type of cancer.23
25
Risk increases with age up to 14 years, with an average age of tumor development between 10 and 11 years.23
26
Early spaying reduces risk. According to one study, intact females are 7 times more likely to develop mammary tumors than cats that are spayed at a young age.27
The latest age at which spaying effectively reduces FMC risk has not been determined.
FMC is most often detected when pet owners or veterinarians feel a mass during examination of the mammary region. However masses can also indicate other conditions, such as severely inflamed lesions, cysts, follicular tumors, and others.4 Histology can be used to confirm the diagnosis and classify the lesion. Tumors are graded as well differentiated, moderately differentiated, or poorly differentiated, based on the appearance of the tissue/cells under a microscope.
Veterinarians and researchers agree that tumor size is the most important prognostic factor in FMC cases, affecting the progression of the disease and the survival time.4 In general, larger tumors have a worse prognosis. Lymph node involvement and degree of metastasis also determine the severity of FMC cases.
Treatment
Surgical removal of mammary tumors is the most common treatment choice. This may include nodulectomy (removal of one or more cell clusters), partial or complete removal of the affected mammary gland(s), removal of lymph nodes, and mastectomy (removal of all glands on the affected side(s)). Veterinarians may use radiation or chemotherapy in addition to surgery. Drugs used to treat FMC include 5-fluoroucil, doxorubicin, cyclophosphamide, methotrexate, prednisone, and vincristine.28
It has been shown that cats that respond to doxorubicin-cyclophosphamide treatments survive for longer periods of time.29
Visit the following sections to learn more:
Cancer in Dogs

Dogs get a variety of cancer types. The most common is skin cancer. Other common canine cancers include breast cancer, bone cancer, and leukemia. Other types of cancer found in dogs include cancer of the mouth, lymphoma, testicular, breast and abdominal tumors.30 Osteosarcoma, or bone cancer, is most common in large dog breeds, such as Great Danes, mastiffs, Labrador Retrievers, and Rottweilers. 3 30 Cancers in dogs are often very similar to those in humans. Genetic risks for canine cancers can be the same as human disease.31
Canine Transmissible Venereal Tumor
Overview and Transmission
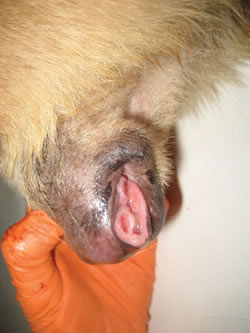
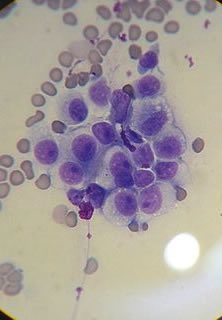
Cancer in humans is practically never transmitted from one person to another. Only very rare documented exceptions exist, usually involving surgical mishaps. Sticker's sarcoma or Canine Transmissible Venereal Tumor (CTVT) is a very unusual form of cancer affecting canines (i.e. dogs). CTVT is transmitted by mating, licking, or other direct contact.32 The tumor affects the genitalia. In some cases the urethra becomes blocked making it difficult for the affected animal to urinate.33 The image below (left) is of a CTVT tumor, showing a typical 'cauliflower-like' mounds. The second image below (right) is a view of CTVT cancer cells as seen in a microscope. If the cancer is located at the mouth and nose, nosebleeds, facial swelling, and nostril discharge are common symptoms.
Many human cancers are caused by viruses, including the human papilloma virus (HPV), a major cause of cervical cancer. Infection with viruses can lead to changes in normal cells within the infected person and lead to the development of cancer. CTVT is different! In this case, the cancer cells themselves are transmitted from animal to animal. Once in the new animal, the tumor can grow and eventually be spread to additional animals.34 Experiments have shown that CTVT is not transmissible via killed tumor cells or by cell contents.32 Viruses are usually present in a cell's liquid contents; if the liquid contents do not transfer CTVT, it is evidence that viruses are not responsible for the cancer. Also, all tumor cells examined so far have a molecular "fingerprint" in their DNA that is absent in normal cells. Specifically, the cancer cells contain a DNA sequence called Line-1 inserted near a known cancer-causing gene called c-myc.35
Researchers have compared tumor DNA and normal DNA within different breeds of dogs. The results showed the expected differences between the normal cell DNA, but all tumor DNA samples were very similar despite being from very different dogs. These results indicate that the tumor cells themselves transfer CTVT between animals.34
The transmission of the cancer depends on physical contact between two dogs. Most of the time, the transmission of the cancer occurs during mating. It is also possible for a dog to get the cancer by sniffing a dog with the cancer. Because male dogs are more likely to sniff strange dogs, they are more likely to get the cancer in this way.36
In most cases, the immune system recognizes and eliminates cells of other types that are introduced into an animal (or human). This does not happen with CTVT. Upon the initial infection, CTVT begins a state of rapid and intense growth that lasts anywhere from three to nine months and possibly longer in old or weakened dogs.32 This is usually followed by a variable regression phase.
Treatment for CTVT
Treatment choices for CTVT include surgery, chemotherapy, or radiation therapy. Chemotherapy is often very effective, usually resulting in complete remission. Surgery does not have a similarly high success rate.37 Because the cancer is transmitted between animals and across generations, the cancer itself is thought to be very old! CTVT is thought to be anywhere from 200-2500 years old and represents the longest line of unbroken replications of a mammal's body cell. Aside from domesticated dogs, it is also transmissible to coyotes, foxes, jackals, wolves and immuno-suppressed mice.32 38
CTVT is not transmissible to humans!
Canine Osteosarcoma
Introduction
Osteosarcoma (OSA) is a cancer that develops within bones. It is often invasive and metastatic.39 OSA is relatively rare in small domesticated animals, accounting for only 5-6% of canine malignancies.40 However, it is by far the most common primary bone tumor in dogs, accounting for 80% of cases.40 41
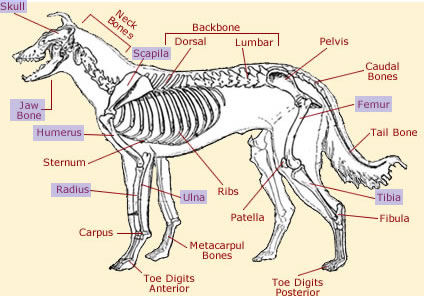
The diagram above highlights some of the bones affected by canine osteosarcoma. This type of cancer occurs most commonly in the appendicular skeleton, including the radius and ulna, femur, tibia, scapila, humerus, and paw bones (phalanges). The front limbs (forelimbs) are more likely to be affected than the rear (hind) limbs. OSA also occurs in the axial skeleton, including the spinal bones, skull, jaw bone (mandible), and the vertebrae at the base of the spine (sacrum). In rare cases, it can affect extraskeletal tissues, such as muscle.40 41 Because of its ability to metastasize, OSA can spread to other parts of the body. Studies have shown that OSA invades the lungs in approximately 17% of cases.41
Risk Factors/Detection/Staging
Osteosarcoma is more common in adult dogs. Large animals, weighing from 44 to 88 lbs (20 to 40 kg), are at a higher risk than smaller animals.41
Signs of OSA in dogs include impaired movement due to pain and stiffness. Affected dogs will often limp to avoid putting weight on the tumor-afflicted bone. They also present with abnormal bone growth and swelling.
Large tumor size is associated with poor prognosis.42 The location of the tumor can also affect the outcome of the disease. Axial skeletal OSA and appendicular OSA tend to have similar prognoses.43 Tumors in the humerus bone tend to have a poor prognosis while tumors in the jaw tend to have better outcomes.42 43
Treatment
Limb amputation is the standard treatment for OSA tumors located in the limbs. Analgesics such as morphine are used to relieve pain, and most dogs can walk within 12-24 hours after the surgery. Many dogs are able to adapt to the amputation within one month. Interestingly, studies have shown that an owner's positive attitude can speed the adaptation process.44 Possible post-surgical complications include infection and recurrence of OSA in the stump.
Certain conditions can make dogs bad candidates for amputation, including arthritis, obesity, neurological problems and large breed. For this reason, vets will perform limb-sparing surgeries, in which they remove the tumor and leave the surrounding tissue and bone intact. This surgery has a higher rate of post-operation infection and OSA recurrence than amputation. Studies reveal that the most common reason vets are asked to perform limb-sparing surgery is not based on the animals condition. Rather, it is that owners are reluctant to proceed with amputation.45 46
Vets can use radiation as a curative treatment or to reduce pain and make the animal more comfortable (palliative care). This type of therapy can reduce inflammation and heal micro-fractures. Studies show that 70% of dogs respond positively to radiation therapy and experience pain relief.40
Chemotherapy drugs such as doxorubicin, carboplatin, cisplatin, or a combination of these drugs may also be used. Chemotherapy alone has not been shown to extend survival time. OSA-affected dogs treated with surgery and chemotherapy tend to survive longer.47
Unfortunately, despite the treatment options available, the prognosis for canine osteosarcoma is poor. The estimated survival time is 6 to 12 months from the time of diagnosis, regardless of the treatment used.39 The most common cause of death (or euthanasia) is disease spread (or metastasis).45
Acknowledgements: The above diagram was found using Creative Commons and can be accessed here. It was obtained and modified on 8/10/09.
Canine Hemangiosarcoma
Introduction
Hemangiosarcoma (HSA) is a cancer that originates in cells that form blood vessels. HSA is responsible for approximately 7% of canine cancer cases. In fact, it is more common in dogs than any other species of animal examined!48
HSA occurs most commonly in the spleen, skin, and the right atrium of the heart.49 Primary tumors have also been reported in the lung, aorta, kidney, oral cavity, muscle, bone, urinary bladder, prostate gland, vagina, lining of the abdominal cavity (peritoneum), intestine, tongue, and the membrane that covers the white part of the eye (conjunctiva).50 51 HSA is highly metastatic, meaning that it frequently spreads. When HSA is confined to the skin, it is less apt to spread. Veterinarians estimate that over 80% of all other cases have already metastasized at the time of diagnosis.52 53
HSA cells may release proteins that stimulate the growth of new blood vessels. This process is known as angiogenesis, and is critical for tumor formation. The presence of numerous blood vessels helps supply growing tumors with nutrients and oxygen and may serve as a "highway" for the cancer cells to metastasize to distant parts of the body.
Risk Factors
Older animals are at higher risk for developing hemangiosarcoma. Affected dogs are usually diagnosed between 9 and 12 years of age.54
HSA tends to affect large breed animals more often than smaller ones. Breeds that are more prone to this type of cancer include German Shepherds, Golden Retrievers, Labrador Retrievers, Pointers, Boxers, English Settlers, Great Danes, Poodles, and Siberian Huskies. Breeds with short hair, light hair, light skin, and/or less pigmentation (i.e. Whippets) are more prone to hemangiosarcoma of the skin55
56
Detection/Symptoms
Symptoms of HSA vary depending on the location and stage of the cancer, but may include weakness, weight loss, increased heart rate (tachycardia), abnormally fast breathing (tachypnea), abdominal swelling, lethargy, and paleness of mucous membranes. Episodes of acute weakness and/or collapse may indicate that a tumor has ruptured, causing uncontrolled blood loss. If internal bleeding occurs and the blood is reabsorbed from the body cavity back into blood vessels, the animal may gradually recover. In more severe cases, tumor rupture can cause sudden death.57
Veterinarians often detect HSA during physical examinations. They also use blood tests, urine analysis, chest x-rays, abdominal ultrasound, ultrasound of the heart (echocardiogram), tissue samples (biopsy), and more. One type of blood test, the complete blood count (CBC), can detect anemia, misshapen or fragmented red blood cells, and other signs of HSA. Some tests can even indicate the organ(s) being affected by the disease. A blood smear that shows an increased number of immature red blood cells (normoblasts) may indicate cancer of the spleen.58 59
Treatment
Surgery is the standard treatment for dogs with HSA. In one study, researchers from Colorado State University attempted surgery on 76 animals with HSA (66 dogs and 10 cats). In 42 cases, surgeons were able to remove the tumor(s) completely, leaving no visible cancer behind. Following surgery, all 42 of these animals remained healthy for at least 12 months. In the remaining 34 cases, the animals experienced incomplete tumor removal and/or other complications, resulting in worse clinical outcomes. These findings confirm that complete tumor removal is the most significant prognostic factor for this type of cancer. Researchers also found that certain tumors (i.e. eye or skin) were much easier to remove than others (i.e. bone or muscle).60
Veterinarians can use other methods to treat and manage hemangiosarcoma, but their effectiveness is limited. Chemotherapy can be used in addition to surgery. Research has shown doxorubicin to be the most effective chemotherapy drug for this type of cancer. Despite treatment, the survival time for HSA-affected dogs typically does not exceed 6 months. Death often results from metastasis to the lungs and other parts of the body.48
Canine Mast Cell Tumors
Introduction
Mast cell tumors, also called mastocytomas, are the most common skin cancer in dogs.61
Mastocytomas develop most often in dogs seven and a half to nine years of age, but can occasionally be found in dogs as young as four to six months.62
Different breeds also have different rates of mast cell tumors. Boxers and Boston terriers have the highest rates.62
Mast cell tumors originate from mast cells, immune system cells found in many tissues of the body.63 Because mast cells can be found almost anywhere, mastocytomas have the potential to appear in all different regions of the body. They are most commonly located on the skin on hind legs, the region between the neck and abdomen (thorax), or genital regions.64 Mast cells contain a variety of biologically active substances, including histamine, heparin, serotonin, and prostaglandins. These chemicals are released from the cells during an allergic reaction.65 These are the substances that cause the symptoms associated with allergies: redness, itching, swelling, blood pressure drops, tearing, nausea, wheezing, ect. Normally these chemicals are highly regulated, but when mast cells become cancerous they no longer appropriately control the release of the chemicals.
Risk Factors/Detection/Staging
The unregulated release of chemicals by a mastocytoma can cause a variety of symptoms. The symptoms any particular dog shows is variable, but can include vomiting, ulcers, bloody excrement, abdominal pain, and blood-clotting difficulties; gastro-intestinal problems are the most common signs.66 Mastocytomas appear as raised lumps that are often irritated or red in appearance. Unfortunately, mast cell tumors have no characteristic form or color and are impossible to identify without laboratory testing.61 Often when a mastocytoma is touched the skin becomes red, itchy, and swollen.62 This response is also called Darier's Sign. It occurs because when the mast cells in the tumor are compressed they release the chemical histamine, and this causes irritation of the skin.
Although normal mast cells are typically fairly stationary, advanced mast cell tumors may spread (metastasize) to other places in the body. Veterinarians classify (stage) mast cell tumors using a six tiered system (stages 0 to 5) based on the extent that the cancer has spread in the body.61 Staging takes into account the number and size of tumors, lymph node involvement, and recurrence rate.61 A higher stage indicates greater body involvement with Stage 5 representing distant metastasis including bone marrow or blood involvement.62 Another classification system, the histological grade (values from 1-3), is based on the physical appearance of the cells in the tumor. More abnormal mast cells/tissues are associated with a higher histological grade and have a higher possibility of being malignant.62
Treatment
Surgical removal of the tumor is the most common treatment for mastocytomas of intermediate and lower stages. Healthy tissue up to about one inch (2-3 centimeters) around the tumor must also be removed to prevent leaving any cancerous mast cells behind. For more advanced stages surgery is still recommended, but is often combined with radiation therapy or chemotherapy.62
If surgery is not thorough enough to remove all cells, external beam radiation therapy can be used to kill the remaining cells. External beam radiation may also be used to reduce the size of a tumor before surgery.62
Cancer in Reptiles

A leopard gecko.
The leopard gecko is reptile. They are one of the more common reptiles to be kept as pets.
A rare type of gecko - a variety known as 'Lemon Frost' (LF), has been shown to spontaneously develop skin cancers that share a defect in common with some human melanomas. The LF gecko has been found to have defects in a gene called SPINT1. The gene is a known tumor suppressor. It encodes a protein that is a serine peptidase inhibitor.67 68
In the gecko, the result of defective SPINT1 genes is the development of skin cancer.69 70 71 The cells affected are involved in the production of skin pigments. In humans, defects in the comparable gene is linked to cancers of pigment producing cells in the skin.
Researchers are now using the LF gecko as a model to help understand the human cancers.
Learn more about tumor suppressors.
If you find the material useful, please consider linking to our website
- 1Urry, L. A., Cain, M. L., Wasserman, S. A., Minorsky, P. V., & Reece, J. B. (2017). Campbell Biology (11th ed.). Pearson.
- 2American Veterinary Medical Association. Material accessed: July 2009. [http://www.avma.org/careforanimals/animatedjourneys/pethealth/pethealth.asp]
- 3 a b c "Cancer Prevalent in Pets but Treatable, Says Veterinarian". Kansas State University. [http://www.newswise.com/articles/view/626941/?sc=dwhn]
- 4 a b c d e Zappulli V, De Zan G, Cardazzo B, Bargelloni L, Castagnaro M. Feline mammary tumors in comparative oncology. 2005 Journal of Dairy Research 72 98-106. [http://journals.cambridge.org/action/displayAbstract?fromPage=online&aid=330933&fulltextType=RA&fileId=S0022029905001263]
- 5National Pet Wellness (2009) How six-month wellness exams can help your pet live a longer, healthier life. [http://www.npwm.com/abt_campaign.htm]
- 6Bertone ER, Snyder LA, Moore AS 2002 Environmental Tobacco Smoke and Risk of Malignant Lymphoma in Pet Cats. American Journal of Epidemiology 156 268-273. [http://aje.oxfordjournals.org/cgi/content/abstract/156/3/268]
- 7 a b OConnor Jr TP, Tonelli QJ, Scarlett JM (1991) Report of the National FeLV/FIV Awareness Project. Journal of the American Veterinary Medical Association 199, 1348-1353. [PUBMED]
- 8Levy JK, Scott HM, Lachtara JL, Crawford PC (2006b) Seroprevalence of feline leukemia virus and feline immunodeficiency virus among cats in North America and risk factors for seropositivity. Journal of the American Veterinary Medical Association 228, 371-376. [PUBMED]
- 9Hardy WD Jr, Hess PW, MacEwen EG, et al. Biology of feline leukemia virus in the natural environment. Cancer Res 1976; 36: 582-588. [PUBMED]
- 10Pacitti AM, Jarret O, Hay D. Transmission of feline leukemia virus in the milk of a nonviraemic cat. Vet Rec 1986; 118: 381-384. [PUBMED]
- 11 a b Levy J, Crawford C et al (2008), 2008 American Association of Feline Practitioners feline retrovirus management and guidelines. Journal of Feline Medicine and Surgery 10: 300-316. [PUBMED]
- 12 a b c d Lutz, H et al (2009) FELINE LEUKEMIA: ABCD guidelines on prevention and management. Journal of Feline Medicine and Surgery 11, 565-574. [PUBMED]
- 13Terry A, Kilbey A, Naseer A, et al. Barriers to Infection of Human Cells by Feline Leukemia Virus: Insights into Resistance to Zoonosis. J Virol. 2017;91(5):e02119-16. Published 2017 Feb 14. doi:10.1128/JVI.02119-16 [PubMed]
- 14Riedel N, Hoover EA et al (1988), Pathogenic and host range determinants of the feline aplastic anemia retrovirus. Proc. Natl. Acad. Sci. 85: 2758-2762. [PUBMED]
- 15Weiss RA, Tailor CS (1995), Retrovirus Receptors. Cell 62: 531-533. [http://www.cell.com/issue?pii=S0092-8674(00)X0394-7]
- 16Cornell University College of Veterinary Medicine (Accessed July 2009) Feline Leukemia Virus [http://www.vet.cornell.edu/fhc/brochures/felv.html]
- 17 a b c d Marretta, J.J., Garrett, L.D., Marretta, S.M. Feline oral squamous cell carcinoma: an overview. Veterinary Medicine, June 2007; p392-406. [http://veterinarymedicine.dvm360.com/vetmed/article/articleDetail.jsp?id=433715]
- 18"Squamous Cell Carcinoma." Long Beach Animal Hospital. Accessed 2 June 2010 [http://www.lbah.com/feline/scc.html#intro]
- 19Ogilvie GK, Moore AS, Obradovich JE, Elmslie RE, al et. Toxicoses and efficacy associated with the administration of mitoxantrone to cats with malignant tumors. J Am Vet Med Assoc, 202: 1839-1844, 1993. [PUBMED]
- 20Ogilvie, Gregory K. "Ten Best Kept Secrets for Treating Cats with Cancer." Proc. of 27 World Small Animal Veterinary Association Congress, Spain, Granada. 2 June 2010 [http://www.vin.com/proceedings/Proceedings.plx?CID=WSAVA2002&PID=2636]
- 21 a b Ferreira, I., S. Rahal, N. Rocha, A.H. Gouveia, T., Y.K. Carvalho, V. Bagnato, Hematoporphyrin-based photodynamic therapy for cutaneous squamous cell carcinoma in cats. Veterinary Dermatology 20(2): 174-178, 2007. [PUBMED]
- 22De Vos, J. P., A.G.O. Burm, B.P. Focker, and De Ottenhorst. "Results from the treatment of advanced stage squamous cell carcinoma of the nasal planum in cats, using a combination of intralesional carboplatin and superficial radiotherapy: a pilot study." Veterinary and Comparative Oncology 2 (2004): 75-81. Wiley InterScience. 17 Aug. 2004. John Wiley & Sons, Inc. 15 July 2009. [http://www3.interscience.wiley.com/journal/118814840/abstract]
- 23 a b c d e Hayes HM Jr, Milne KL, Mandell CP. Epidemiological features of feline mammary carcinoma. Vet Rec. 1981;108(22):476–479. doi:10.1136/vr.108.22.476 [PUBMED]
- 24Misdorp W, Romijn A and Hart AA 1991 Feline mammary tumors: a case-control study of hormonal factors. Anticancer Research 11 1793-1797.Misdorp W, Romijn A, Hart AA. Feline mammary tumors: a case-control study of hormonal factors. Anticancer Res. 1991;11(5):1793–1797. [PUBMED]
- 25Ito T, Kadosawa T, Mochizuki M, Matsunaga S, Nishimura R, Sasaki N. Prognosis of malignant mammary tumor in 53 cats. J Vet Med Sci. 1996;58(8):723–726. doi:10.1292/jvms.58.723 [PUBMED]
- 26Misdorp W, Romijn A and Hart AA 1991 Feline mammary tumors: a case-control study of hormonal factors. Anticancer Research 11 1793-1797. [PUBMED]
- 27Weijer K, Hart AA. Prognostic factors in feline mammary carcinoma. J Natl Cancer Inst. 1983;70(4):709–716. [PUBMED]
- 28Stolwijk JA, Minke JM, Rutteman GR, Hoekstra J, Prop FJ, Misdorp W. Feline mammary carcinomas as a model for human breast cancer. II. Comparison of in vivo and in vitro adriamycin sensitivity. Anticancer Res. 1989;9(4):1045–1048. [PUBMED]
- 29Stolwijk JA, Minke JM, Rutteman GR, Hoekstra J, Prop FJ, Misdorp W. Feline mammary carcinomas as a model for human breast cancer. II. Comparison of in vivo and in vitro adriamycin sensitivity. Anticancer Res. 1989;9(4):1045–1048. [PUBMED]
- 30 a b Heather L. Gardner, Joelle M. Fenger and Cheryl A. London. Dogs as a Model for Cancer. 2016. Annu Rev Anim Biosci. 2016; 4: 199–222. Published online 2015 Nov 9. doi: 10.1146/annurev-animal-022114-110911 [ href=“https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6314649/“ target=“_blank”>PubMed]
- 31. (2020). Mutations of BRCA2 in canine mammary tumors and their targeting potential in clinical therapy. Bmc Veterinary Research, 16(1), 30. http://doi.org/10.1186/s12917-020-2247-4 (Original work published January 2020) [PUBMED]
- 32 a b c d Cohen, D. (1985). The canine transmissible venereal tumor: a unique result of tumor progression. Adv. Cancer Res. 43, 75112. [PUBMED]
- 33Hasler A, Weber W (2000). "Theriogenology question of the month. Transmissible venereal tumor (TVT)." J. Am. Vet. Med. Assoc. 216 (10): 15579. [PUBMED]
- 34 a b Murgia C, Pritchard JK, Kim SY, Fassati A, Weiss RA. "Clonal Origin and Evolution of a Transmissible Cancer". Cell (2006) 126(3):47787. doi:10.1016/j.cell.2006.05.051. [PUBMED]
- 35Katzir N, Rechavi G, Cohen JB, Unger T, Simoni F, Segal S, Cohen D, Givol D. "Retroposon" insertion into the cellular oncogene c-myc in canine transmissible venereal tumor. Proc. Natl. Acad. Sci. USA (1985) 82, 10541058. [PUBMED]
- 36Male dogs are more likely to develop contagious nose cancer from sniffing other dogs’ genitals. The Conversation. Published July 4, 2022. Beth Daley, Editor https://theconversation.com/male-dogs-are-more-likely-to-develop-contagious-nose-cancer-from-sniffing-other-dogs-genitals-186259 Accessed 08-03-2022
- 37Ettinger, Stephen J; Feldman, Edward C. (1995). Textbook of Veterinary Internal Medicine (4th ed.). W.B. Saunders Company. ISBN 0-7216-6795-3. [http://en.wikipedia.org/w/index.php?title=Special%3ABookSources&isbn=0721667953]
- 38Harmelin A, Pinthus JH, Katzir N, Kapon A, Volcani Y, Amariglio EN, Rehavi G. (2001). "Use of a murine xenograft model for canine transmissible venereal tumor." Am. J. Vet. Res. 62, 907911. [PUBMED]
- 39 a b Thompson JP, Fugent MJ. Evaluation of survival times after limb amputation, with and without subsequent administration of cisplatin, for treatment of appendicular osteosarcoma in dogs: 30 cases (1979-1990). Journal of the American Veterinary Medical Association (1992) 200(4): 531-533. [PUBMED]
- 40 a b c d Endicott M. Principles of treatment for osteosarcoma. Clin Tech Small Anim Pract (2003) 12(2): 110-114 [PUBMED]
- 41 a b c d Cavalcanti JN, Amstalden EMI, Guerra JL, Magna LC. Osteosarcoma in dogs: clinical-morphological study and prognostic correlation. Brazilian Journal of Veterinary Research and Animal Science (2004) 41: 299-305. [http://www.scielo.br/scielo.php?pid=S1413-95962004000500002&script=sci_arttext]
- 42 a b Kuntz CA, Asselin TI, Dernell WS, et al. Limb salvage surgery for osteosarcoma of the proximal humerus: Outcome in 17 dogs. Vet Surg (1998) 27: 417-422. [PUBMED]
- 43 a b Straw AC, Powers BE, Kalusner J, et. al. Canine mandibular osteosarcoma: 51 cases (1980-1992). J Am Anim Hosp Assoc (1996) 32: 257-263. [PUBMED]
- 44Kirpensteijn J, van den Bos R, Endenburg N. Adaption of dogs to the amputation of a limb and their owners satisfaction with the procedure. Vet Rec(1999) 144: 115118. [PUBMED]
- 45 a b Dernell WS, Straw RC,Withrow SJ. Tumors of the skeletal system in Withrow SJ, MacEwen EG (eds): Small Animal Clinical Oncology (2001) pp 378-417. [http://books.google.com/books?id=wprLBe_0U5QC&pg=PA1&lpg=PA410&dq=(7)%09Dernell+WS,+Straw+RC,Withrow+SJ.+%E2%80%9CTumors+of+the+skeletal+system%E2%80%9D&source=bl&ots=r7lhpEbD4G&sig=gjm3XGCGvaZEgnqIldi_-4OJDDI&hl=en&ei=bpSBSqvzLOG_tgfw2KTSCg&sa=X&oi=book_result&ct=result&resnum=4#v=onepage&q=(7)%09Dernell%20WS%2C%20Straw%20RC%2CWithrow%20SJ.%20%E2%80%9CTumors%20of%20the%20skeletal%20system%E2%80%9D&f=false]
- 46Straw RC, Withrow SJ. Limb-sparing surgery versus amputation for dogs with bone tumors. Vet Clin North Am Small Anim Pract (1996) 26: 135143. [PUBMED]
- 47Liptak JM, et. al. Canine Appendicular Osteosarcoma: Curative-Intent Treatment. Compendium on Continuing Education for the Practicing Veterinarian (March 2004) pp. 186-197. [http://animalcancersurgeon.com/Review_Articles_files/Compendium%202004%20OSA%20II%20curative-intent.PDF]
- 48 a b Helfand SC. Canine hemangiosarcoma: A tumor of contemporary interest. Cancer Therapy (2008) 6: 457-462.
- 49Pearson GR, Head KW. Malignant haemangioendothelioma (angiosarcoma) in the dog. Journal of Small Animal Practice (1976) 17: 737-745. [PUBMED]
- 50. (2017). Actionable mutations in canine hemangiosarcoma. Plos One, 12(11), e0188667. http://doi.org/10.1371/journal.pone.0188667 (Original work published December 2017) [PUBMED]
- 51. (2018). Canine sarcomas as a surrogate for the human disease. Pharmacology & Therapeutics, 188, 80-96. http://doi.org/10.1016/j.pharmthera.2018.01.012 (Original work published December 2018) [PUBMED]
- 52. (2017). Actionable mutations in canine hemangiosarcoma. Plos One, 12(11), e0188667. http://doi.org/10.1371/journal.pone.0188667 (Original work published December 2017) [PUBMED]
- 53. (2018). Canine sarcomas as a surrogate for the human disease. Pharmacology & Therapeutics, 188, 80-96. http://doi.org/10.1016/j.pharmthera.2018.01.012 (Original work published December 2018) [PUBMED]
- 54Bertazzolo W, et. al. Canine angiosarcoma: cytologic, histologic, and immunohistochemical correlations. Veterinary Clinical Pathology (2005) 34(1): 28-34. [PUBMED]
- 55Ward H, Fox LE, Calderwood-Mays MB, Hammer AS, Cuoto CG. Cutaneous hemangiosarcoma in 25 dogs: a retrospective study. J Vet Intern Med (1994) 8: 345-348. [PUBMED]
- 56Hargis AM, Ihrke PJ, Spangler WL, Stannard AA. A retrospective clinicopathologic study of 212 dogs with cutaneous hemangiomas and hemangiosarcomas. Vet Pathol (1992) 1992: 316-328. [PUBMED]
- 57. (2018). Canine sarcomas as a surrogate for the human disease. Pharmacology & Therapeutics, 188, 80-96. http://doi.org/10.1016/j.pharmthera.2018.01.012 (Original work published December 2018) [PUBMED]
- 58. (2017). Actionable mutations in canine hemangiosarcoma. Plos One, 12(11), e0188667. http://doi.org/10.1371/journal.pone.0188667 (Original work published December 2017) [PUBMED]
- 59. (2018). Canine sarcomas as a surrogate for the human disease. Pharmacology & Therapeutics, 188, 80-96. http://doi.org/10.1016/j.pharmthera.2018.01.012 (Original work published December 2018) [PUBMED]
- 60Schultheiss PC. A retrospective study of visceral and nonvisceral hemangiosarcoma and hemangiomas in domestic animals. J Vet Diagn Invest (2004) 16: 522-526. [PUBMED]
- 61 a b c d Nash, DVM, MS, Holly. "Canine Mast Cell Tumors: A Common Cancer in Dogs." Dog, Cat, and Pet Care Tips, Health and Behavior Information by Veterinarians. Veterinary Services Department. Web. 2 June 2010. [http://www.peteducation.com/article.cfm?c=2+1638&aid=461]
- 62 a b c d e f g Welle MM, Bley CR, Howard J, Rufenacht S. Canine mast cell tumours: a review of the pathogenesis, clinical features, pathology and treatment. Veterinary Dermatology (2008) 19(6): 321-39. http://www3.interscience.wiley.com/cgi-bin/fulltext/121451889/HTMLSTART [http://www3.interscience.wiley.com/cgi-bin/fulltext/121451889/HTMLSTART] [PUBMED]
- 63"Mast Cell." Encyclopædia Britannica. 2009. Encyclopædia Britannica Online. 2 June 2010 [http://www.britannica.com/EBchecked/topic/368641/mast-cell]
- 64"Cutaneous Mast Cell Tumors." The Merck Veterinary Manual. 2008. [http://www.merckvetmanual.com/mvm/index.jsp?cfile=htm/bc/72231.htm&word=canine%2cmast%2ccell]
- 65. (2019). Mast Cell Biology at Molecular Level: a Comprehensive Review. Clinical Reviews In Allergy & Immunology. http://doi.org/10.1007/s12016-019-08769-2 (Original work published December 2019) [PUBMED]
- 66Scott MA, Stockham SL: Basophils and mast cells. Schalm's Veterinary Hematology, 5th ed. Feldman BF, Zinkl JG, Jain NC (eds). Lippincott Williams & Wilkins, 2000, pp. 308-315.
- 67Wilcox, Christie. “Lemon Frost” Leopard Geckos’ Cancers Similar to Human Melanomas. The Scientist. June 24, 2021. https://www.the-scientist.com/news-opinion/lemon-frost-leopard-geckos-cancers-similar-to-human-melanomas-68924
- 68Gene Card: https://www.genecards.org/cgi-bin/carddisp.pl?gene=SPINT1 Accessed 08-10-2021
- 69. (2020). Iridophoroma associated with the Lemon Frost colour morph of the leopard gecko (Eublepharis macularius). Scientific Reports, 10(1), 5734. http://doi.org/10.1038/s41598-020-62828-9 (Original work published December 2020) [PUBMED]
- 70. (2021). Author Correction: Iridophoroma associated with the Lemon Frost colour morph of the leopard gecko (Eublepharis macularius). Scientific Reports, 11(1), 7248. http://doi.org/10.1038/s41598-021-87009-0 (Original work published March 2021) [PUBMED]
- 71. (2021). Genetics of white color and iridophoroma in "Lemon Frost" leopard geckos. Plos Genetics, 17(6), e1009580. http://doi.org/10.1371/journal.pgen.1009580 (Original work published June 2021) [PUBMED]